Nucleotide Metabolism
Nucleotide Metabolism. Dr. S.Chakravarty , MD. A 30 year old man comes with severe pain in great toe of right foot !!. Learning objectives. Discuss the components of Nucleotides and molecules contributing to formation of purine and pyrimidine ring

Nucleotide Metabolism
E N D
Presentation Transcript
Nucleotide Metabolism Dr. S.Chakravarty, MD
A 30 year old man comes with severe pain in great toe of right foot !!
Learning objectives • Discuss the components of Nucleotides and molecules contributing to formation of purine and pyrimidine ring • Describe the Pyrimidine metabolism and its defects – Oroticacidurias and Megaloblasticanemias • Differentiate the features of Denovoand salvage pathways of purine metabolism • List the causes of Hyperuricemias, its clinical features and treatment • Discuss the clinical features and the enzyme defects in SCID and Lesch - Nyhan syndrome – clinical features • List the anticancer drugs acting on purine and pyrimidine metabolism and its mechanism of action
Importance of nucleotides: • Building blocks of nucleic acids – DNA and RNA • Act as co-enzymes – FAD, NAD, NADP • Second messengers – cAMP and cGMP • Energy currency – ATP and GTP • Nucleoside and nucleotide analogs – treatment of cancer. • Donors of sugar moiety – UDP-Glucuronic acid.
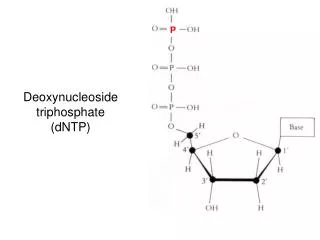
Nomenclature • Base= (Purines or Pyrimidines) • Nucleosides = Base + sugar • Nucleotides = Base + sugar + phosphate • Sugar = Ribose in RNA = 2 - Deoxyribose in DNA
Pyrimidines Mnemonic : CUT
Pyrimidine biosynthesis • The donors of C and N atoms to the pyrimidine ring are Aspartate, Glutamine and CO2 • The pyrimidine ring is initially assembled & the ribose-phosphate is added later (compare to purine biosynthesis)
De BIOSYNTHESIS OF PYRIMIDINES From Kaplan Step 1 lecture notes
CO2 + ATP Glutamine -VE OROTIC ACIDURIA -VE -VE Hydroxyurea Dihydrofolatereductase -VE -VE 5-flurouracil Methotrexate – Eukaryotes Trimethoprim – prokaryotes Pyrimethamine - protozoal
Conversion of the ribonucleotides to deoxyribonucleotides Ribonucleotidediphosphate (ADP, GDP, CDP, UDP) Thioredoxin, NADPH+H+ Ribonucleotidereductase (-) Hydroxyurea Deoxyribonucleotidediphosphate (dADP, dGDP, dCDP, dUDP)
Carbomyl phosphate synthase • Carbamoyl phosphate synthetase I • - involved in urea synthesis • - uses free ammoniaas the source of nitrogen • - occurs in liver mitochondria • - activated by N-acetylglutamate • - not affected by UDP or UTP • Carbamoyl phosphate synthetase II • - involved in pyrimidine synthesis • - uses glutamine as the source of nitrogen • - occurs in the cytosol of all nucleated cells • - Inhibited by UDP and UTP, Activated by PRPP, ATP • - not activated by N-acetylglutamate
Oroticaciduria • Deficiency in UMP synthase activity • OrotatePhosphoribosyl transferase • Orotidylatedecarboxylase • Pyrimidine synthesis is decreased, excess orotic acid is excreted in urine (hence Oroticaciduria) • Due to demand for nucleotides in red blood cell synthesis megaloblastic anemia • Important :- Unresponsive to vit B12 and folic acid,
Oroticaciduria • Administration of Uridine improves anemia (Uridine is used for the synthesis of thymidine and cytidine) • UTP is feedback inhibitor of CPS II, uridine administration results in a fall in orotic acid levels.
Oroticaciduria – other causes: • Urea cycle defect : Ornithinetranscarbomylase (OTC) • Drugs – Allopurinol competes with orotic acid for the enzyme orotatephosphoribosyl transferase .
Question • A one year old female child is weak and anemic. The child was found to have megaloblastic anemia. The height and weight of the child are less than normal. Urine demonstrates an elevated level of orotic acid excretion and normal blood ammonia levels. • Which of the following enzyme will be deficient ? • A. Ornithinetranscarbamoylase • B. OrotatePhosphoribosyltransferase
Pyrimidine catabolism • Dephosphorylated to nucleotide • Cytosine converted to uracil • Uracil converted to b-alanine • Thymine converted to b-aminoisobutyrate
Purine Biosynthesis N10-Formyl Tetrahydrofolate
Purine synthesis- De novo pathway Allopurinol 6- Mercaptopurine Azathioprine Ribose- 5 phosphate PRPP synthase PRPP (-) (+) (-) PRPP Amidotransferase 5-phosphoribosylamine (-) IMP (Hypoxanthine base) Aspartate Glutamine AMP GMP
Purine catabolism Adenosine Severe combined Immunodeficiency ADENOSINE DEAMINASE NH3 (-) Guanosine T-cell deficiency Insosine (-) (-) Purine nucleoside phosphorylase Purine nucleoside phosphorylase Ribose 1 P Ribose 1 P Hypoxanthine Guanine Guanase Xanthine Oxidase Xanthine (-)Allopurinol Xanthine Oxidase Uric acid Urine
90% 10% From Kaplan Step 1 lecture notes
Purine salvage pathway Inosine Guanine (90%) Adenine Hypoxanthine (90%) PRPP Adenine phosphoribosyl transferase Hypoxanthine Guanine phosphoribosyl transferase PRPP AMP GMP IMP (-) LeschNyhan syndrome • Purines from Diet • Nucleic acid turnover • Other nucleotide metabolism
Advantage of purine salvage pathway: • Reutilization of nucleotides • Prevents loss of ATPs which are required for denovopurine synthesis • Nucleotides formed in the salvage pathway inhibits denovo pathway at the rate limiting step • Decreases uric acid formation – end product of purine catabolism
Conversion of nucleosides to nucleotides • NMP + ATP NDP • NDP + ATP NTP • NDP dNDP (RNA) (DNA) Nucleoside Mono phosphate kinase Nucleoside DI phosphate kinase Conversion of Ribonucleotides to De-oxy Ribonucleotides: Ribonucletidereductase
LeschNyhan syndrome • X-linked recessive disease • Complete deficiency of HGPRT – Hypoxanthine guanine phosphoribosyl transferase. • Increased uric acid production – pathogenesis: • Deficient salvage of nucleosides • Decreased IMP and GMP and increased PRPP- REMOVAL OF FEEDBACK INHIBITON which stimulates PRPP amido transferase – DeNovopurine synthesis
Clinical features • Severe heritable form of Gout – due to increased uric acid production. • Features of Gout. • Self mutilation – head banging, biting of lips and fingers • Involuntary movements- choreoathetotic movements. • Uric acid stones.
Adenosine deaminase deficiency • Part of SCID – severe combined immuno deficiency syndrome. • Autosomal recessive – def of enzyme Adenosine deaminase • Def leads to accumulation of deoxyadenosine which increases the concentration d-ATP. This inturn inhibits ribonucleotidereductasewhich is required for converting ribonucleotides to de-oxyribonucleotides.
Clinical features • Deficiency of both T and B lymphocytes • Leads to deficiency of both cell mediated and humoral immunity. • Prone to infections. • Bubble boy – sterile environment. • Treatment – gene therapy.
Normal URIC ACID LEVEL2-7mg/dl • HYPERURICEMIA is not GOUT !! • HYPERURICEMIA PREDISPOSES TO GOUT !!
GOUT (Podagra) • Recurrent attacks of inflammatory arthritis • Metatarso- phalengeal joint of Big toe – MC • Also presents as tophi, kidneystones and urate nephropathy. • Elevation of uric acid levels – over production or under excretion of uric acid.
Monosodium urate crystals • Poor solubility and hypersaturation leads to formation of needle shaped monosodium urate crystals (negatively birefringent) which are deposited in joints, tendons, and subcutaneous tissues. • crystals attracts inflammatory cells – inflammatory mediators - inflammation
Causes of primary Gout • Decreased excretion : Defect in tubular secretion in kidneys. • Increased production :PRPP synthase mutation – low Km for ribose – 5- PO4 or high Vmax for PRPP production. • HGPRT deficiency- LeschNyhan syndrome
Secondary GOUT • Lactic acidosis • Von-Gierke’s disease • Cancer chemotherapy • Tumorlysis syndrome • Hereditary Fructose intolerance • Renal failure • Purine rich foods.
Treatment of Gout • Colchicine – inhibits migration of inflammatory cells • NSAIDS – Antiiflammatory and analgesics • Uricosuric drugs –probencid and sulfinpyrazone • CONTRAINDICATED IN RENAL GOUT • Xanthine oxidase inhibitors – Allopurinol.
MCQ1 Gout is characterized by elevated uric acid concentrations in blood and urine due to a variety of metabolic abnormalities that lead to the overproduction of purine nucleotides. Allopurinol is used in the treatment of gout because this drug, and its metabolic product, alloxanthine, act as inhibitors of: • a) XanthineOxidase • b) PRPP synthetase • c) Adenylsuccinate synthase • d) Hypoxhantine guanine phosphoribosyltransferase • e) Nucleotides
MCQ2 • Hereditary Oroticaciduria is characterized by severe anemia, growth retardation, and high levels of orotic acid excretion. It is produced by deficit of enzymes related with: • a) synthesis of pyrimidine nucleotides • b) catabolism of pyrimidine nucleotides • c) synthesis of purine nucleotides • d) catabolism of purine nucleotides • e) synthesis of Heme • f) catabolism of Heme