Half-Life
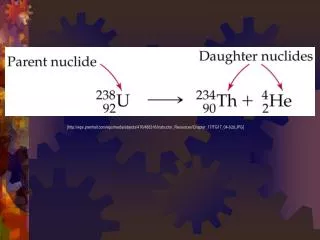
Half-Life . Definition . A HALF-LIFE is the amount of TIME it takes for HALF of a radioactive sample to decay. Half-Life is found on Table N (Get out your Reference Tables!). Seriously . …get out your reference tables!. Two amounts to be concerned with:.


Half-Life
E N D
Presentation Transcript
Definition • A HALF-LIFE is the amount of TIME it takes for HALF of a radioactive sample to decay. • Half-Life is found on Table N (Get out your Reference Tables!)
Seriously • …get out your reference tables!
Two amounts to be concerned with: • TIME: Every half-life is the same unit of time (That same value on Table N) • MASS: The remaining mass is cut in HALF each half-life. • If you go BACKWARDS in time, the mass DOUBLES!
Example 1.: • How many grams of a 64.0 gram sample of Ca-37 remains undecayed after 728 ms?
Example 1. • From Table N: Calcium-37 has a half-life of 182 ms. Starting at TIME = ZERO, we have 64.0 grams. Every 182 ms we cut the remaining amount in half, until we reach the time given in the problem (728 ms) • It’s a good idea to setup the following table: