Half-Life
Half-Life. Pg. 870-874. Half-Life. Half-life = the time needed for half of the radioisotope sample to decay into products Half-live can have varying lengths May be a fraction of a second Can last billions of years. Examples: Carbon-14 = 5730 years Potassium-40 = 1.25x10 9 years

Half-Life
E N D
Presentation Transcript
Half-Life Pg. 870-874
Half-Life • Half-life= the time needed for half of the radioisotope sample to decay into products • Half-live can have varying lengths • May be a fraction of a second • Can last billions of years • Examples: • Carbon-14 = 5730 years • Potassium-40 = 1.25x109 years • Radon-22 = 3.8 days • Thorium-230 = 75,400 years • Thorium-234 = 24.1 days
Calculating Half lives • There are 2 ways to solve these problems: • Continually divide by 2 • Use the equation below N = No (1/2) n
Calculating Half-Life • Nitrogen-13 emits beta radiation and decays to carbon-13 with a half-life (t1/2) of 10 min. Assume a starting mass of 2.00g of nitrogen-13. • How long is three half-lives? • How many grams of the isotope will be present at the end of three half-lives?
Calculating with Half lives • If the passing of 5 half lives leaves 25.0mg of strontium-90 sample, how much was present in the beginning?
Examples of Uses of Half-Life asymmetric scan indicates disease normal • Can be used to determine the age of an artifact (carbon-14 dating) • Artificial isotopes tend to have short have lives - Used in medicine - Don’t pose long-term hazards to patients
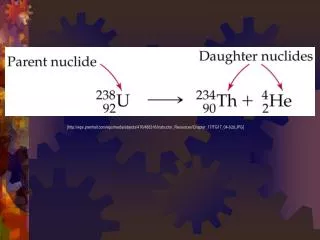
Radiocarbon dating • AKA: Carbon-14 dating • Carbon-14 is produced in the atmosphere when high-energy neutrons from space (cosmic rays) collide with nitrogen-14 • Carbon-14 decays by b-particle production
Carbon Dating • Carbon-14 is continuously produced in the atmosphere and it continuously decomposes by β-decay • The two opposite processes have come into balance, so the amount of carbon-14 present in the atmosphere is approximately constant.
Carbon Dating • Carbon-14 dating can be used to date wood and cloth artifacts • A living plant consumes carbon dioxide in the photosynthesis process • Some carbon dioxide contains the C-14 isotope • When the plant uses carbon dioxide with C-14, it is incorporated into the molecules in the plant
Carbon Dating • While it is alive, the C-14 in the plant molecules is equal to that in the atmosphere. • When the tree is cut, there is no more incoming source of C-14 to balance the C-14 that is decaying. • The C-14 content decreases.
Carbon Dating • Using the known half-life of 5730 years for C-14, archaeologists compare the amount of C-14 in an artifact to the amount currently in trees and figure out the age. • A wooden bowl with half as much C-14 as a current living tree would have been through one half life and would therefore be 5730 years old. Carbon dating made by Thomas Athol Rafter
Half life Activity • You must get the data today • Class data will be posted on Moodle tonight • Lab due Monday