9- Nucleotide Metabolism
250 likes | 378 Vues
Explore roles of nucleotides in metabolism: genetic material, energy currency, biosynthesis carriers, coenzymes, and more. Discover de novo and salvage pathways, purine synthesis, regulation, degradation, and clinical disorders in purine metabolism.

9- Nucleotide Metabolism
E N D
Presentation Transcript
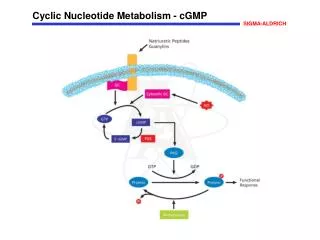
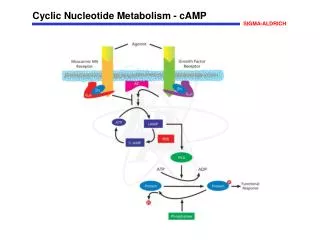
Roles of Nucleotides 1. Precursors to nucleic acids (genetic material and non-protein enzymes). 2. Currency in energy metabolism (eg. ATP, GTP). 3. Carriers of activated metabolites for biosynthesis (eg. CDP, UDP). 4. Structural moieties of coenzymes (eg. NAD, CoA). 5. Metabolic regulators and signal molecules (eg. cAMP, cGMP, ppGpp).
Biosynthetic routes: De novo and salvage pathways • De novo pathways • Almost all cell types have the ability to synthesize purine and pyrimidine nucleotides from low molecular weight precursors in amounts sufficient for their own needs. • The de novo pathways are almost identical in all organisms.
Salvage pathways • Most organisms have the ability to synthesize nucleotides from nucleosides or bases that become available through the diet or from degradation of nucleic acids. • In animals, the extracellular hydrolysis of ingested nucleic acids represents the major route by which bases become available.
Reutilization and catabolism of purine and pyrimidine bases blue-catabolism red-salvage pathways endonucleases: pancreatic RNAse pancreatic DNAse phosphodiesterases: usually non-specific
De novo Synthesis of Purine Nucleotides • Atoms of purine ring are contributed by a number of compounds, including amino acids (aspartic acid, glycine, and glutamine), CO2, and N10–formyltetrahydrofolate. • The purine ring is constructed by a series of reactions that add the donated carbons and nitrogens to a preformed ribose 5-phosphate.
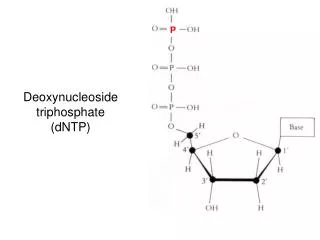
Synthesis of 5-phosphoribosyl-1-pyrophosphate (PRPP) • PRPP is an “activated pentose” that participates in the synthesis and salvage of purines and pyrimidines. • Synthesis of PRPP from ATP and ribose 5-phosphate is catalyzed by PRPP synthetase (ribose phosphate pyrophosphokinase).
Example of a salvage pathway: guanine phosphoribosyl transferase In vivo, the reaction is driven to the right by the action of pyrophosphatase.
Synthesis of IMP (inosine monophosphate “parent purine nucleotide” The base in IMP is called hypoxanthine Note: purine ring built up at nucleotide level. precursors: glutamine (twice) glycine N10-formyl-THF (twice) HCO3 aspartate In vertebrates, 2,3,5 catalyzed by trifunctional enzyme, 6,7 catalyzed by bifunctional enzyme.
Pathways from IMP to AMP and GMP G-1: IMP dehydrogenase G-2: XMP aminase A-1: adenylosuccinate synthetase A-2: adenylosuccinate lyase Note: GTP used to make AMP, ATP used to make GMP. Also, feedback inhibition by AMP and GMP.
Pathways from AMP and GMP to ATP and GTP Conversion to diphosphate involves specific kinases: GMP + ATP <-------> GDP + ADP Guanylate kinase AMP + ATP <-------> 2 ADP Adenylate kinase Conversion to triphosphate by Nucleoside diphosphate kinase (NDK): GDP + ATP <------> GTP + ADP NDK also works with pyrimidine nucleotides.
Synthetic inhibitors of purine synthesis • Some synthetic inhibitors of purine synthesis (for example, the sulfonamides), are designed to inhibit the growth of rapidly dividing microorganisms without interfering with human cell functions. • Other inhibitors, such as structural analogs of folic acid (for example, methotrexate), are used pharmacologically to control the spread of cancer by interfering with the synthesis of nucleotides and, therefore, of DNA and RNA. • Trimethoprim, another folate analog, has potent antibacterial activity because of its selective inhibition of bacterial dihydrofolate reductase.
Inhibitors of human purine synthesis are extremely toxic to tissues, especially to developing structures such as in a fetus, • or to cell types that normally replicate rapidly, including those of bone marrow, skin, gastrointestinal (GI) tract, immune system, or hair follicles. • As a result, individuals taking such anticancer drugs can experience adverse effects, including anemia, scaly skin, GI tract disturbance, immunodeficiencies, and baldness.
Purine degradation AMP deamination in muscle, hydrolysis in other tissues. Xanthine oxidase:contains FAD, molybdenum, and non-heme iron. In primates, uric acid is the end product, which is excreted.
Clinical disorders of purine metabolism Excessive accumulation of uric acid: Gout The three defects shown each result in elevated de novo purine biosynthesis
Common treatment for gout: allopurinol Allopurinolis an analogue of hypoxanthine that strongly inhibits xanthine oxidase . Xanthine and hypoxanthine, which are soluble, are accumulated and excreted.
Diseases of purine metabolism (continued) Lesch-Nyhan Syndrome: Severe HGPRT deficiency In addition to symptoms of gout, patients display severe behavioral disorders, learning disorder, aggressiveness and hostility, including self-directed. Reason for the behavioral disorder is unknown. X-linked trait (HGPRT gene is on X chromosome). Severe combined immune deficiency (SCID): lack of adenosine deaminase (ADA). Lack of ADA causes accumulation of deoxyadenosine. Immune cells, which have potent salvage pathways, accumulate dATP, which blocks production of other dNTPs by its action on ribonucleotide reductase. Immune cells can’t replicate their DNA, and thus can’t mount an immune response.
Pyrimidine Synthesis and Degradation • Pyrimidine ring is synthesized before being attached to ribose 5-phosphate, which is donated by PRPP. • The sources of the atoms in the pyrimidine ring are glutamine, CO2, and aspartic acid.
Formation of a pyrimidine nucleotide • The completed pyrimidine ring is converted to the nucleotide orotidine 5′-monophosphate (OMP) in the second stage of pyrimidine nucleotide synthesis. • OMP, the parent pyrimidine mononucleotide, is converted to uridine monophosphate (UMP) by orotidylate decarboxylase, which removes the acidic carboxyl group.
Orotic aciduria—a rare genetic defect—is caused by a deficiency of the Orotate phosphoribosyltransferase and orotidylate decarboxylase (a bifunctional enzyme), resulting in orotic acid in the urine. • UMP is phosphorylated to UTP.
Salvage of pyrimidines • Few pyrimidine bases are salvaged in human cells. • However, the pyrimidine nucleosides can be salvaged by nucleoside kinases that utilize ATP in the phosphorylation of the nucleosides to nucleotides.
Degradation of pyrimidine nucleotides • Unlike the purine ring, which is not cleaved by human cells, the pyrimidine ring is opened and degraded to highly soluble products, β-alanine and β-aminoisobutyrate, with the production of NH3 and CO2.