Salts
Salts. Salt Solutions. The salts of weak acids can recombine with water producing basic solutions. Salt Solutions. The salts of weak bases can recombine with water producing acidic solutions. Salt Solutions. A - + H 2 O HA + OH - B + + H 2 O H + + BOH.

Salts
E N D
Presentation Transcript
Salt Solutions • The salts of weak acids can recombine with water producing basic solutions
Salt Solutions • The salts of weak bases can recombine with water producing acidic solutions
Salt Solutions • A- + H2O HA + OH- • B+ + H2O H+ + BOH
Salt Problem • Calculate the pH of a 0.20 M solution of NaBz • Ka = 6.4 x 10-5
Salt Problem • Calculate the pH of a 0.10 M solution of R-NH3Cl • Kb = 2.5 x 10-5
Drill: Calculate the pH of a 0.18 M solution of KC2H3O2 • Ka = 1.8 x 10-5
Drill: Calculate the pH of a 0.16 M solution of KC7H5O2 • Ka = 6.4 x 10-5
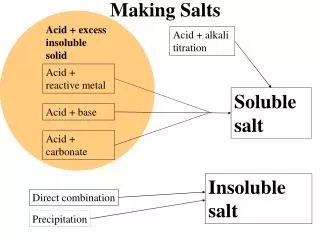
Salt Applications • Salts of strong acids & weak bases make acidic solutions
Salt Applications • Salts of strong bases & weak acids make basic solutions
Salt Applications • Salts of strong acids & strong bases make neutral solutions
Predict Relative pH • NaAc BaCl2 • KNO3 NH4Br • KHSO4 NH4Ac
Predict Relative pH • KAc NaCl • KClO2 NH4Cl • K2SO4 NaI
Anhydrides • Compounds without water; that when added to water, form other compounds
Acid Anhydrides • Non-metal oxides that form acids when added to water
Basic Anhydrides • Metal oxides that form bases when added to water
Predict Relative pH • Na2O SO2 • NO2 CO2 • CaO Al2O3
7.4 g of Ca(OH)2 was added to 2.0 L of 0.075 M H2CO3. Calculate the molarity of all ions present in the solution.Ka1 = 4.4 x 10-7Ka2 = 4.7 x 10-11
Calculate the pH of 0.10 M HF in 0.20 M NaF. Ka HF = 6.5 x 10-4
Calculate [H3PO4], [H2PO4-1], [HPO4-2], [PO4-3], [K+], [H+], & pH of 1.0 M KH2PO4 in 0.50 M K2HPO4. Ka1 = 7.5 x 10-3Ka2 = 6.2 x 10-8Ka3 = 4.2 x 10-13
Calculate pH of: 0.50 M MOHin 0.20 M MCl Kb = 5.0 x 10-5
Calculate pH of: 0.20 M MCl Kb = 5.0 x 10-5
Review of Acid/Base descriptions and Acid/Base, Buffer, & Salt Equilibria
Strong Acids Strong Bases
Acid rxns Base rxns
A/B Equilibrium Constants KW, KA, KB, & pH
Calculate pH of: • 0.025 M HNO3 • 0.020 M KOH
Calculate pH of: • 2.0 M HNO2 • Ka = 2.0 x 10-4
Calculate pH of: • 3.0 M HZin • 2.0 M KZ • Ka HZ = 3.0 x 10-5
Calculate pH of: • 0.20 M KQ • Ka HQ = 2.0 x 10-5
150 mL of 0.10 M KOH was added to 100.0 mL of 0.10 M H2CO3. Calculate [H2CO3], [HCO3-], [CO3-2], [H+], [OH-], & pH of the solution.Ka1 = 4.4 x 10-7Ka2 = 4.7 x 10-11
Calculate [H3A], [H2A-1], [HA-2], [A-3], [K+], [H+],[OH-], & pH of 2.0 M KH2A. Ka1 = 4.0 x 10-3Ka2 = 5.0 x 10-8Ka3 = 2.5 x 10-13
Calculate pH of: • 2.0 M HQ • Ka = 2.0 x 10-6
Calculate pH of: • 6.0 M HZin • 4.0 M KZ • Ka HZ = 3.0 x 10-5
Calculate pH of: • 0.20 M KQ • Ka HQ = 2.0 x 10-7