Structure of the atom
Structure of the atom. Amlan Ganguly Lecturer, Dept. of Pharmacy NUB. Rutherford’s atom model :

Structure of the atom
E N D
Presentation Transcript
Structure of the atom Amlan Ganguly Lecturer, Dept. of Pharmacy NUB
Rutherford’s atom model: The atom model that Rutherford proposed very much confirms our solar system. For this reason it is called Rutherford’s solar system atom model. The major propositions of the model are:
1.An atom consists of a small nucleus containing all the positive charges of the atom and practically the whole of its mass. 2. The nucleus is surrounded by a number of electrons equal to the number of positive charge on the nucleus. 3. The electrons move in orbits or closed paths around the nucleus like planets move around the sun. 4.The electrostatic force of attraction between the electrons and positive nucleus is counterbalanced by the outward centrifugal force produced by the motion of electrons.
Limitations of Rutherford’s atom model Rutherford’s atom model is based upon Newton’s Laws of motion and gravitation. Scientist Bohr pointed out some limitations of Rutherford’s atom model which are as follows: 1. Newton’s Laws of motion and gravitation can only be applied to neutral bodies such as planets and not to charged bodies such as tiny electrons moving round a positive nucleus. The analogy does not hold good since the electrons in an atom repel one another, whereas planets attract each other because of gravitational forces. Besides, there is electrostatic attraction in a nuclear atom model.
2. According to Maxwell’s theory, any charged body such as electrons rotating in an orbit must radiate energy continuously. If an electron radiates energy, its speed will decrease and it must gradually spiral in towards the nucleus. The radius of the electron will gradually decrease and it will ultimately fall into the nucleus, thus annihilating the atom model. This does not happen actually. This was the chief weakness of Rutherford’s Atomic Model. An electron revolving round the nucleus in orbit continuously emits radiations and loses energy, and ultimately falls into the nucleus. 3. Rutherford did not give any idea about shape or size of the pattern in which the electrons move around the nucleus.
Bohr’s atom model • In 1913 Niels Bohr constructed a theory of hydrogen atom in particular and to some extent of atomic structure in general. Bohr’s theory was based on Planck’s quantum theory and was built on the following postulates: • 1. Postulate concerning energy levels: An atom has a number of stable circular orbits in which an electron can revolve without the radiation of energy. These orbits are referred to as “ Energy levels”. The stationary orbits depending on their distance from the nucleus are stated as the 1st, 2nd, or 3rd energy level. As the distance from the nucleus increases, their energy increases. The electrons in these orbits possess an integral multiple of the quantum of energy i.e., hʋ, but do not radiate it.
Representation of various orbits (energy levels) round the nucleus. E1. E2. E3 ….etc. are the energies associated with orbits having n=1 (K-shell), 2 (L-shell), 3 (M-shell)…..etc. Energies of different orbits are in the order: E1< E2< E3…..etc 2. Postulate concerning angular momentum: Ifthe electron moves in a circular orbit, its motion is subject to the ordinary laws of electrical and centrifugal forces. These orbits are decided by the condition that the angular momentum of the electron in such orbits must be an integral multiple of h/2π, that is, The angular momentum, mvr= n.(h/2π) Where, m is the mass of the electron, v is the velocity, r is the radius of the orbit( distance of an electron from the nucleus), h is the Planck’s constant and n is 1,2,3,4,5 etc. The quantity n is called the principal quantum number. It also represents the serial number of allowed orbits in order of increasing distance from the nucleus.
3.Postulate concerning radiation of energy: The atoms emit or absorb energy when the electron jumps from one orbit to another. An atom radiates energy as light only when an electron passes from a higher energy level to another of lower energy givingdefinite spectral lines in emission spectra. The jump of an electron from a lower energy level to that of a higher energy level is manifested by absorption of energy giving dark lines. The quantity of energy radiated or absorbed is equal to the difference between the energies the atom possessed before and after the electron changed orbits. If E1 and E2 are the energies of the electron in the initial and final levels respectively, the difference of energy radiated when the electron passes from the higher to the lower energy level is given by the relation. E2-E1= hʋ Where h is the Planck’s constant and ʋ is the frequency of radiation.
Limitations of Bohr’s atomic model • No explanation of fine structure: Bohr’s theory had successfully explained the origin of hydrogen spectrum. But when the hydrogen spectrum is observed by means of spectroscope of high resolving power, it is found that individual spectral lines are not really single but consist of several very fine lines lying close together. This fine structure of spectral lines in the hydrogen spectrum cannot be explained by Bohr’s theory. • 2. No explanation for the spectra of multi-electron system: Bohr’s theory has successfully explained the origin of the spectrum of hydrogen atom which is one-electron system, but it is not able to explain the origin of the spectrum of multi-electron systems.
3. No explanation for Zeeman effect: Ithasbeen observed that when an excited atom which gives a line emission spectrum is put in magnetic field, its spectral lines are further split up into still thinner (i.e. closely-spaced) lines. This type of splitting of spectral lines into a number of closely spaced lines is called Zeeman effect. Bohr’s theory gives no explanation for Zeeman effect.4.According to Bohr’s model electrons revolve in circular orbits but modern research reveals that electrons may revolve in elliptical orbits also. • 5. Byprecisely defining the radius of the atomic orbits, the theory violated Heisenberg’s uncertainty principle according to which it is impossible to define simultaneously the position and momentum of a moving sub-atomic particle. Δx. ΔP= h/2π
Δx= error or uncertainty in the measurement of the position of a moving subatomic particle • Δp= error or uncertainty in the measurement of the momentum of a moving subatomic particle. • Sommerfield’s modification of Bohr’s atomic model: • Sommerfield modified Bohr’s theory in 1915 to explain the fine structure in the spectrum of hydrogen atom. According to this modification the electrons in any particular energy level could have either a circular orbit or a number of elliptical orbits about the nucleus. • Only those elliptical orbits are selected in which the electron possesses the angular momentum given by the equation, • Mvr= h√l(l+1)/2π, where l stands for subsidiary or azimuthal or orbital quantum number.
Thomson’s Atomic Model (1893) • An atom can be regarded as a heavy positively charged sphere which consists of a positively charged substance distributed uniformly throughout the whole sphere. • The electrons are in the sphere as the seeds are embedded in a water melon. • The mutual repulsion existing between different electrons keeps them charged in the form of circular shells. • The atomic model was given up after some time since it could not explain the large deflection suffered by α-particles in Rutherford experiment.
Rutherford’s experiment on the scattering of α particles by thin metal foil • Narrow beam of α –particles from radioactive • substances like radium placed in a lead block • ZnS screen helped in detecting the direction • in which α –particles moved. • When α –particles struck the screen, a flash • of light is observed. • Most of the particles passed through the foil • without any deflection from the path and • struck the screen at the centre, causing • illumination. • A few of them were deflected at some angle • (90° or wider angle) • Very few of them were deflected at the same • turn back on their original path (not more than • 1 in 10,000)
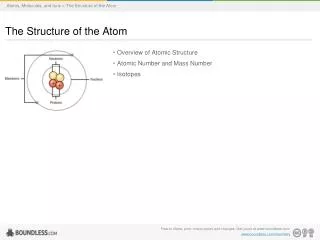
Rutherford’s Nuclear Atomic Model • Atom contains a massive (heavy) and positively charged part at its centre. This central part of atom is called nucleus. • The volume occupied by the nucleus is only a minute fraction of total volume of atom, i.e. the size of the nuclueus is very small as compared to the whole atom • Atom is not at all solid, as earlier suggested by Dalton, but is extraordinarily hollow, since consists of a lot of empty space round the nucleus. • Electron are revolving round the nucleus in closed orbit with a fast speed like solar system.
Quantum number: • The numbers, which express the size, shapes, direction of the orbital from the nucleus and the spins of the electron of their own axis are called quantum numbers. • There are four types of quantum numbers. • 1. Principal quantum number • 2. Subsidiary quantum number or Azimuthal quantum number • 3. Magnetic quantum number • 4. Spin quantum number
Principal quantum number : • This expresses the orbits or principal energy levels to which the electron belongs. It is denoted by n ( n=1, 2, 3 etc). For the first orbit, n=1, for the second orbit, n=2 and so on. • The energy levels are also called electron shells. Inner most shell is known as K shell and the succeeding outer shells are denoted by the letter L, M, N, O, P, Q etc. • Principal quantum number represents any particular circular orbit. The value of n gives roughly the binding force and the distance between the nucleus and the electron and also the energy it possesses. The first energy level is the one nearest the nucleus. Consequently, it is strongly bound with nucleus and possesses the lowest energy. • The quantum number also represents the size of the electron orbit.
2. Subsidiary quantum number or Azimuthal quantum number: • The main energy levels (or shells) of electron may be considered as being made up of one or more sub- levels (sub-shells). The number, which is used to express the sub-level in which the electron belongs, is known as subsidiary quantum number or Azimuthal quantum number. It is denoted by l. • The value of l depends on the value of n (Principal quantum number). For a given value of n, l can have all integral values from 0 to n-1 each of which refers to an energy sublevel or sub-shell. • The values of l i.e. the subsidiary quantum number 0,1,2,3, are also symbolized by letters s, p, d and f respectively. • Subsidiary quantum numbers describe the shapes of the electron orbit energy level. The orbits are circular when l= n-1 and have elliptical shapes when l is different from n-1.
3. Magnetic quantum number/ Orientation quantum number: • The magnetic quantum number was introduced to explain the orientation of electronic orbit in space particularly under the influence of an applied magnetic field. This is designated by m. • The permitted values of m are dependent upon l. m can have values from –l to +l including 0 i.e.,. There are (2l+1) values. • For l=1 ( p orbital), the magnetic quantum number m will have three values: +1. 0 and -1. So there are three orientations for p orbital. These three types of p orbital differ only in the value of magnetic quantum number and are designated as Px, Py, Pz depending upon the axis of orientation. The subscripts x, y and z refer to the co-ordinate axis.
4. Spin quantum number: Thespin quantum number represents the direction of the electron spin and is designated by s. • The direction of spin is investigated in presence of external magnetic field and it is found that all electrons spin either in one direction (say, clockwise direction) or in the direction directly opposite (anti-clockwise). Their values are, s=+1/2 and s=- ½. They are often represented by putting two arrows in the opposite directions. ↑and ↓
Pauli’s Exclusion Principle: • Pauli’s exclusion principle states that, no two electrons in the same atom can have the same values for all four quantum numbers. • The above rule appears to be analogous to the law: Two bodies cannot be placed in the same position at the same time”. Thus, two electrons of the same atom can have the same values for three of their quantum numbers, but the fourth quantum number must be different for the two electrons. Thus two electrons may have orbits of the same size, shape and orientation in space provided they have opposed spins.
Aufbau’s law: • Electrons go to the different orbital according to their increasing energy. Electrons first go to the orbital of low energy and then systematically to the orbital of higher energy. It is known as Aufbau’s laws. • The orbital which has lower value of (n+l) has the lower energy and higher value of (n+l) has higher energy. • Here, n= principal energy level • L= subsidiary energy level • Example: Considering 4s and 3d orbital: • For the 4s orbital, the value of, n=4 and the value of l=0 (s). The total value of (n+l)= 4+0= 4 • Again for the 3d orbital, the value of, n=3 and the value of l=2 (d). The total value of (n+l)= 3+2= 5 • So, electrons will go to 4s orbital first between 4s and 3d orbital.
Considering 3d and 4p orbital: • For the 3d orbital, the value of n=3 and the value of l=2 (d). The total value of (n+l) = 3+2=5 • For the 4p orbital, the value of n=4 and the value of l=1(p). The total value of (n+l) = 4+1=5 • If the two orbital have the same value of (n+l) then electrons will go to that orbital first, which has lower principal quantum number. So electrons will go to the 3d first.
Distribution of electrons in the atoms of electrons: • Theelectronsarearranged among the known elements in seven main energy levels designated by the principal quantum numbers n=1,2,3,4,5,67. These principal levels are divided into sub-levels indicated by s,p,d, and f. • The first energy level (n=1) which has only one sub-level is designated as 1s. The second energy level (n=2) has two sub-levels designated as 2s and 2p, the third (n=3) energy level has three sub-levels designated as 3s, 3p and 3d and the fourth energy level n=4 has four sub-levels 4s, 4p, 4d and 4f. • The sub-levels are further divisible into orbitals. An s sub-level is made up of one orbital; a p sub-level three -orbitals; a d sub-level five- orbitals and f sub-level- seven orbitals.
Each electron orbital can accommodate a maximum of two electrons of opposed spins. Thus 1s orbital can hold a maximum of two electrons. The three p orbitals can hold a maximum of 6 electrons etc. • Thus we get each electron shell can hold a maximum of 2n2electrons where n is the shell number.
Spectrum • A spectrum is an array of waves or particles which is spread out according to the increasing or decreasing of some property such as wavelength or frequency. • Types of spectra: Depending on the nature of the source emitting the radiation there are two types of spectra. • (1) Emission spectra • (2) Absorption spectra
Spectra Absorption spectra Emission spectrum Continuous spectrum Discontinuous spectrum Line spectrum (atomic spectrum) Band spectrum (molecular spectrum)
(1) Emission Spectra: Emission spectra can be obtained from the substances which emit light on excitation. Their excitation can be done as follows: • 1) By heating the liquid or solid substances in a flame at high temperature. These substances become incandescent at high temperature. When these substances are heated at high temperature, they emit light. • 2) By passing an electric discharge through a gas at low pressure. • 1 (a)Continuous spectrum: • When a narrow beam of sunlight or any white light (e.g. an arc lamp) is allowed to pass through a prism, different wavelengths are refracted (or bent) through different angles. When received on a screen, these form a series of color bands viz., violet, indigo, blue, green, yellow, orange and red (VIBGYOR)
The series of bands that form a continuous rainbow of colors on the screen is called a continuous spectrum. Each of the seven colors of the spectrum has got a characteristic wavelength, lie in range of 4000-7500 A0 • In the spectrum, one color merges into another without break or discontinuity, that’s why, the spectrum thus obtained is called continuous spectrum. • 1(b) Discontinuous spectrum: • (i) Line spectrum or atomic spectrum: • When an atom of an element absorbs energy, it gets excited. The excited atom emits light radiations of a characteristic colour. When this emitted radiation is allowed to pass through a prism, it is resolved into individual components or bright lines. • Such a spectrum in which each line represents a specific wavelength of radiation emitted by the atoms is referred to as the line spectrum. Sincethis spectrum is characteristic of
of atoms, it is also called atomic spectrum. • This kind of spectrum is characteristic of the atom of the element and hence is used to identify the atom in a material. • Examples of atomic spectrum: Spectrum given by Na, K, Li atoms in their excited state and that given by hydrogen gas filled in a discharge tube at reduced pressure are the important examples of atomic spectra.
(ii) Band spectrum or molecular spectrum: • Band spectrum is the property of molecules and generally given by the compounds or gases like N2 and O2 molecules at low temperature and pressure. The band spectrum consists of a number of bright bands, separated by dark spaces. Since this spectrum is characteristic of molecules, it is also called molecular spectrum. Each band is sharp at one end and fades gradually as we approach the other end.
(2) Absorption spectrum: • When white light, composed of all visible wavelengths, is passed through a substance, certain wavelengths may be absorbed. These absorbed wavelengths are thus found missing in the transmitted light. The spectrum obtained in this way consists of a series of dark lines in a particular region of the continuous spectrum of white light which is referred to as absorption spectrum. • For example, if a solution of NaCl is placed between the sun light and prism, the continuous spectrum of white light is found to be crossed by two dark lines in the yellow region of the spectrum. • If the white light source is removed, the whole of the continuous spectrum disappears leaving behind only dark yellow lines. These dark lines are known as absorption line spectrum.
Atomic emission spectrum of hydrogen: • When an electric discharge is passed through hydrogen gas filled in a discharge tube at a very low pressure, a bluish light is emitted from the discharge tube. When the ray of this emitted bluish light is passed through a prism, a discontinuous line spectrum consisting of several isolated bright lines with dark area in between is obtained. Such a spectrum is called line spectrum of hydrogen. When an electric discharge is passed through hydrogen, the molecules of hydrogen break into atoms. These atoms absorb energy from the electric spark and get excited. The excited atoms give the extra energy in the form of emitted light (bluish light)
Bohr’s explanation of hydrogen spectrum: • The solitary electron in hydrogen atom at ordinary temperature resides in the first orbit (n=1) and is the lowest energy state (ground state). When energy is supplied to hydrogen gas in the discharge tube, the electron moves to higher energy levels viz., 2,3,4,5,6,7, etc., depending on the quantity of energy absorbed. From these high energy levels, the electron returns by jumping to one or other lower energy level. In doing so, the electron emits the excess energy as light radiation of some definite frequencies which will give rise to emission spectra. This gives an excellent explanation of the various spectral series of hydrogen.
Lyman series is obtained when the electron returns to the ground state i.e., n=1 from higher energy levels (n2 = 2,3,4,5,etc). Similarly, Balmer, Paschen, Brakett and Pfund series are produced when the electron returns to the second, third, fourth and fifth energy levels respectively . Classification of spectral lines in hydrogen spectrum: • Scientist Balmer observed that there were four prominent coloured lines in the visible region of hydrogen spectrum. • (i) a red line with a wavelength of 6563 A • (ii) a blue-green line with a wavelength of 4861 A • (iii) a blue line with a wavelength of 4340 A • (iv) a violet line with a wavelength of 4102 A • The above series of four lines in the visible spectrum of hydrogen was named as the Balmer Series.
In addition to Balmer series, four other spectral lines were discovered in the infrared and ultraviolet regions of the hydrogen spectrum which are named after the names of their discoverers. Thus all these spectral lines can be classified into five groups or series:
Rydberg’s equation: • Bymeasuring the wavelength of a large number of lines observed in different spectral series of hydrogen spectrum, Rydberg gave an empirical equation with the help of which the frequency of any line of a given series of hydrogen spectrum can be calculated. This equation can be written as: ʋ- =R (1/n12-1/n22) Where, ʋ- = frequency in terms of wave number ( in cm-1 or m-1) λ= wavelength R= a universal constant which is called Rydberg’s constant. Its experimental value is 109678 cm-1 n1 = represents the lower energy level where the electron returns from the higher energy level. It remains constant for a given series.
n2 = represents the higher energy level where the electron returns to the lower energy level. • Wave number (ʋ- ): In spectroscopy , frequency of light is generally expressed in terms of wave number, which is defined as the number of wavelengths per meter (or per centimeter) and is equal to the reciprocal of wavelength (λ). • It is denoted by (ʋ- ) and Thus: • Wave number (ʋ- ) = 1/ λ ʋ- is expressed in cm-1 or m-1
Calculate the wavelength of light emitted from H for the electronic transition from energy level 4 to 2 using R= 109676 cm-1 • Solution: • The wavelengths of lines in Balmer series are given by • 1/ λ= R (1/n12-1/n22) • Where, λ= wavelength, R (Rydberg constant) = 109,676 cm-1 • n1 =2 and n2= 4 • 1/ λ= 109676 ( 1/ 22 -1/ 42 ) • 1/ λ= 109676 × (3/16) • λ= 16/ (109676 × 3) • Λ= 4.86 × 10-5 cm • Wavelength of the spectral line is = 4.86 × 10-5 cm
Self practice: • A spectral line in the Lyman series of hydrogen spectrum has a frequency of 82,200cm-1. What transition is responsible for this radiation? ( R= 109,600cm -1) • Calculate the frequency of spectral lines emitted in case of hydrogen atom when the electron jumps from level 2 to level 1.
Zeeman effect: • It has been observed that when an excited atom which gives a line emission spectrum is put in magnetic field, its spectral lines are further split up into still thinner ( i.e. closely-spaced ) lines. This type of splitting of spectral lines into a number of closely spaced lines is called Zeeman effect. Bohr’s theory gives no explanation for Zeeman effect. It is assumed that the secondary energy levels are further sub-divided into other energy levels by the presence of magnetic field. These subdivisions of the secondary energy levels are called orbitals. • A schematic diagram shown below gives the resolutions of the energy levels to show the splitting of the spectral lines. It is to be noted that s energy levels are not splitted in a magnetic field, whereas p type secondary energy levels appear to consist of three orbitals.
Mosley’s law: • When cathode rays are allowed to fall on a metal target called anti-cathode or target material placed in their path, new type of radiations are produced. These radiations are called X-rays. The stream of cathode rays (i.e. electrons ) produced from the cathode converges to a point through a concave surface on the anti-cathode and gives rise to X-rays. • Mosley found that when cathode rays struck different elements used as target materials in the discharge tube, characteristic X-rays were emitted. The wavelength of these X-rays decrease in a regular manner in passing from one element to the next one in order in the Periodic table. • Mosley plotted the atomic number against the square root of the frequency of the X-rays emitted and obtained a straight line which indicated that atomic number was not a mere ‘position number’ but a fundamental property of the atom.
Finally, Mosley gave an expression relating the square root of the frequency (√ʋ) of a characteristic X-ray spectrum and the atomic number (Z) of the element used as anti-cathode (target material) which is known as Mosley’s law. It can be stated as : The square root of frequency of a spectral line on the characteristic X-ray spectrum of an element (used as a target material ) varies directly as the atomic number of the element producing the spectrum, i.e. • √ʋ α Z
Structureofatom • Fundamental particles of atom: • An atom is the smallest particles of an element having its own chemical identity and properties. • Atom consists of still smaller particles, such as electron, proton, neutron etc. into which it can be subdivided. These particles are regarded as fundamental particles, because these are the main constituents of atoms. • Classification of fundamental particles: • i) Permanent fundamental particles: The particles which play an important role for the formation of structure of an atom and are present in all atoms (except hydrogen where neutron is absent) are called permanent fundamental particles. There are three kinds of permanenent fundamen
tal particles such as electron, proton and neutron. • Electron bears negative charge and revolves around the nucleus in a circular path. The mass of electron is very small. • Proton bears the positive charge and stays at the nucleus with neutrons. • Neutron has no charge, i.e. it is neutral. It is placed in the nucleus with proton.
2) Temporary fundamental particles: • The particles which exist in some atoms for short time are called temporary fundamental particles. There are about 100 numbers of temporary fundamental particles in different atom like positron, neutrino, antineutrino, meson etc. • Production of cathode rays (discovery of electron): • The knowledge about the electron was derived as a result of the electric discharge in a discharge tube. The discharge tube consists of a glass tube with metal electrodes fused in the walls. The pressure inside the tube can be reduced with a pump. When the electrodes are connected to a source of high voltage (10,000 volts), an electric discharge from high potential source is passed through a gas at 0.001 mm pressure of Hg in the discharge tube and the glass wall opposite to the cathode
begins to glow with a very faint greenish light. This glow or fluorescence is formed due to the bombardment on the glass by certain rays which are emitted from the cathode –surface and move towards the anode with tremendous velocity. These rays were called cathode rays by Goldstein since these originated from the cathode.
Production of positive rays (discovery of proton): • In 1886, German physicist , Eugen Goldstein used a discharge tube provided with a cathode perforated with extremely fine holes and observed that when the electrodes are connected to a source of high voltage (10,000 volts), not only cathode rays of electrons were originated from the cathode moving from cathode towards anode but also new type of colored rays were simultaneously originated from the anode moving from anode towards cathode through the fine holes or canals in the cathode causing a glow on the wall opposite to anode. • Initially these rays were called canal rays, since they pass through the canals or holes of the cathode. These are also called anode rays as they seem to originate from the anode. Thomson has shown that these rays consist of particles carrying a positive charge and hence he has called them positive rays.
Anode (Positive) Rays-Discovery of Proton • When large potential difference applied to electrode • Not only cathode rays originated • But also new type of colored rays were also simultaneously originated from anode • Moving from anode toward cathode through the fine holes or canals in the cathode causing a glow on the wall opposite to anode • Initially, Canal rays: as they pass through canals or holes • Anode rays: as they originate from anode • J J Thomson proved that these rays are positively charged and hence they are called positive rays 1886 German physicist E. Goldstrein
Explanation of the Production of Positive Rays • When a high voltage is applied across the metallic electrodes of a discharge tube filled with a gas under reduced pressure, the molecules of the gas are dissociated into atoms. • Now when high speed electrons (i.e. cathode rays) originated from cathode strike these atoms, the atoms are converted into cations which pass through the holes of the cathode and constitute positive rays. • The electrons obtained in the conversion of the gaseous atoms into cations move towards anode as negative rays M + e- M+ + 2e- Move towards anode as negative rays Pass through the perforated cathode as positive rays Originated from cathode Gaseous atoms