Structure of the Atom
340 likes | 525 Vues
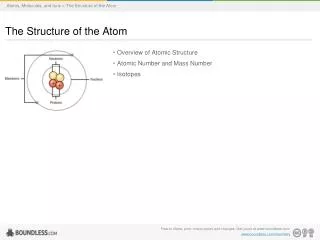
Structure of the Atom. Structure of the atom. Atoms Protons Neutron Electron Nucleus. Historical Background. Continuous theory of Matter: a solid body can be divided and subdivided into smaller pieces without limits.

Structure of the Atom
E N D
Presentation Transcript
Structure of the atom • Atoms • Protons • Neutron • Electron • Nucleus
Historical Background • Continuous theory of Matter: a solid body can be divided and subdivided into smaller pieces without limits. • Discontinuous theory of Matter: particles so small and indestructible ie Democritus and Leucippus
Continued • Dalton’s Atomic Theory • 1. All elements are composed of atoms, which are indivisible and indestructible particles • 2. All atoms of the same element are exactly alike
Dalton’s theory cont • 3. Atoms of different elements are different • 4. Compounds are formed by the joining of atoms of two or more elements. In any compound the atoms of the different elements in the compound are joined in a definite whole number ratio
Law of Conservation of Mass • Originated with Antoine Lavoisier • States that matter can be neither created nor destroyed
Law of Definite Proportions • Determined by Joseph Proust • That the proportion by mass of the elements in a given compound is always the same • For example in water the mass of oxygen is always 8 times the mass of the hydrogen
Law of Multiple Proportions • Certain pairs of elements can combine to form one and only one compound (NaCl) • Masses of one element that combine with a fixed mass of the other element form simple whole-number ratios
Discovery of the Electron • William Crookes- high voltage to a crookes tube and that these cathode rays are deflected by magnetic field • JJ Thompson- deflected by a fundamental particles in a tube called electrons
Discovery of Proton • Rutherford-Gold foil experiments found that something in the nucleus caused deflections and he named it protons • Discovered alpha particle, beta rays and atomic structure for nuclear physics
Energy levels • Bohr-definite orbits in which the electrons encircle the nucleus • Define ground states of electrons are most stable • Define excited states of the electrons is energetically unstable
Energy levels cont • 7 energy levels within an atom • K is the lowest then L M N O P Q all called shells • This is equal to energy levels 1-7
Neutrons • James Chadwick discovered the neutron • Same mass as a proton but has no electrical charge
Models of the atoms • They range from Dalton’s up to the quantum mechanical model. Which basically states that light has a dual nature it can behave both as waves and as particles.
Basic vocabulary to know • Atomic number * Orbitals • Isotopes *Alpha particles • Planck’s constant * Quanta • Wavelength *Atomic mass • Frequency *Mass number • Wave velocity * Atomic mass unit
When light is a wave has properties of • Wavelength=Lamda • Frequency = hz or f • Wave velocity = v = f lambda
Electromagnetic spectrum • Speed of light = 3.00 X 108 m/s • C = speed of light • Includes visable light, IR UV radio x-rays gamma radar microwaves • Frequency increases the wavelength decreases on the scale
Light as energy • Planks constant = h • H = 6.6 X 10 -34 joule/hz • E= hf is the formula • C= f/lambda
Quantum Numbers:four numbers used to describe the electrons in an atom.
Schrodingerdescribed an atomic model with electrons in three dimensions. This model required three coordinates, or three quantum numbers, to describe where electrons could be found.
The three coordinates from Schr–dinger's wave equations are the principal (n), angular (l), and magnetic (m) quantum numbers. These quantum numbers describe the size, shape, and orientation in space of the orbitals on an atom.
1. Principal (shell) quantum number - n • Describes the energy level within the atom. • Energy levels are 1 to 7 • Maximum number of electrons in n is 2n2
2. Momentum (subshell) quantum number - l • Describes the sublevel in n • Sublevels in the atoms of the known elements are s - p - d - f • Each energy level has n sublevels. • Sublevels of different energy levels may have overlapping energies.
The momentum quantum number also describes the shape of the orbital. • Orbitals have shapes that are best described as spherical (l = 0), polar (l = 1), or cloverleaf (l = 2). • Orbitals even take on more complex shapes as the value of the angular quantum number becomes larger.
3. Magnetic quantum number - m • Describes the orbital within a sublevel
s has 1 orbital • p has 3 orbitals • d has 5 orbitals • f has 7 orbitals • Orbitals contain 1 or 2 electrons, never more.
malso describes the direction, or orientation in space for the orbital. • This diagram shows the three possible orientations of a p orbital - px, py, pz. Atomic orbitals are studied in great detail in a college "theoretical inorganic chemistry" class. If you are curious, study these websites
4. Spin quantum number -s • This fourth quantum number describes the spin of the electron. • Electrons in the same orbital must have opposite spins. • Possible spins are clockwise or counterclockwise.
Rules governing the combinations of quantum numbers: • Three quantum numbersn, l, and mare integers. • The principal quantum number (n) cannot be zero. • n must be 1, 2, 3, etc.
The angular quantum number (l) can be any integer between 0 and n - 1. • For n = 3, l can be either 0, 1, or 2.
The magnetic quantum number (m) can be any integer between -l and +l. • For l = 2, m can be either -2, -1, 0, +1, or +2. • The spin quantum number (s) is arbitrarily assigned the numbers +1/2 and -1/2.
Pauli Exclusion Principle: • No two electrons in an atom have the same set of four quantum numbers.
Hund's Rule: • Electrons will enter empty orbitals of equal energy, when they are available.
Quantum Chemistry: Describes the way atoms combine to form molecules and the way molecules interact with one another, using the rules of quantum physics.