Early Theories of Matter: From Democritus to Rutherford's Atom Model
Explore the evolution of atomic theory from ancient Greek philosopher Democritus, who proposed that matter is made up of indivisible particles called "atomos," to Aristotle's rejection of this idea. Discover how John Dalton revived atomic concepts with his own theory in the 19th century, and learn about J.J. Thomson's discovery of the electron, leading to the Plum-Pudding Model, and Ernest Rutherford's groundbreaking experiments that led to the nuclear model of the atom. This lecture delves into the key figures and their contributions to our understanding of atomic structure.

Early Theories of Matter: From Democritus to Rutherford's Atom Model
E N D
Presentation Transcript
Structure of the Atom Lecture 1
Early Theories of Matter • Democritus (460-370 B.C.) – Greek Philosopher • Proposed that matter was not infinitely divisible • Matter was made up of tiny individual particles called atomos • Atomos could not be created, destroyed, or further divided • Different kinds of atomos had different sizes and shapes • Changes in matter resulted from the change in the groupings of atomos and not from the atomos themselves.
Aristotle (384-322 B.C.) – Greek Philosopher • Rejected Democritus’ atomic theory because it did not agree with his own ideas on nature • He did not believe that the “nothingness” of empty space through which atomos moved could exist
John Dalton (1766-1844) • Revived and revised Democritus’ ideas based upon his own scientific research. • Devised his own atomic theory which included • All matter is composed of extremely small particles called atoms • All atoms of a given element are identical, having the same size, mass, and chemical properties • Atoms of a specific element are different from those of any other element • Atoms cannot be created, divided into smaller particles, or destroyed • Different atoms combine in simple whole-number ratios to form compounds • In a chemical reaction, atoms are separated, combined, or rearranged.
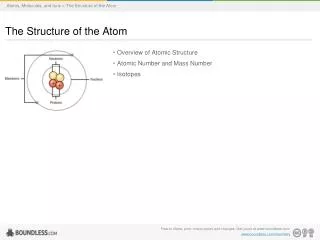
Atoms: Defined • An atom is the smallest particle of an element that retains the properties of the element. • Atoms are composed of subatomic particles found in the nucleus of the atom and outside the nucleus. • Protons: positively charged, mass of 1 amu, found in the nucleus • Neutrons, no charge, mas of 1 amu, found in the nucleus • Electrons: negatively charged, mass of 1/1840thamu, found outside the nucleus in electron valences
Discovering the Electron • J.J. Thomson (1890s) conducted cathode ray (streams of negatively charged particles) experiments to determine the ratio of the charged cathode ray particles to their mass. • He concluded that the mass of the particle was less than that of a hydrogen atom. • He proposed that the atom was composed of a uniform positive charge that had negatively charged electrons distributed throughout. It was called the Plum-pudding model. • Robert Millikan (1909) determined the mass of an electron to be 9.1 x 10-28 g or 1/1840th the mass of a hydrogen atom.
Nuclear Atom • Ernest Rutherford (1911) conducted an experiment to determine how positively charge alpha particles interacted with solid matter. • Alpha particles have the mass of a Helium atom and contain 2 protons and 2 neutrons, but no electrons. • He suspected only minor deflections of the particles based on the Plum-pudding model. He believed that the alpha particles would shoot through matter without interacting with the matter’s atoms. • The alpha particles were sharply deflected back toward the source of the alpha particles or at sharp angles away from the matter.
Rutherford concluded that the atom was composed of a tiny, dense region, he called the nucleus, which contained all of an atom’s positive charge and mass surrounded by mostly empty space containing the electrons. • He hypothesized that electrons were held in place orbiting the nucleus by their attraction to its positive charge. • By 1920, Rutherford concluded that the nucleus contained positively charge particles he called protons. • Protons have a mas of 1.673 x 10-24 g or 1 amu.
James Chadwick (1932) showed that the nucleus also contained another subatomic particle, a neutral particle he called the neutron. • A neutron has a mass nearly equal to that of a proton (1.675x10-24 g or 1 amu).
Atomic Number • Henry Mosely (1887-1915) discovered that each element contained a unique positive charge in its nucleus. • The number of protons identifies each particular element. • The number of protons is the element’s atomic number. • All atoms are neutral and the number of protons must equal the number of electrons.
Isotopes and Mass Number • Atoms with differing numbers of neutrons are called isotopes. • Isotopes have the same number of protons and eletrons, just differing numbers of neutrons. • The mass number of an element is the AVERAGE of the sum of the protons and neutrons in the nucleus of all the isotopes of that element. • Isotopes are given the elemental name and the mass number for that specific isotope. • For example: potassium with 19 protons and 20 neutrons would be Potassium-39 or K-39.
Radioactivity • Changes that involve the nucleus of an atom are referred to as nuclear reactions. • Some atoms spontaneously emit radiation in a process called radioactivity. • Radioactive atoms undergo significant changes that can alter their identities. • By emitting radiation, atoms of one element transform into atoms of a different element.
Radioactive Decay • Radioactive elements emit radiation because their nuclei are unstable. • This process is called radioactive decay. • Atoms undergo radioactive decay until they become stable non-radioactive atoms, resulting in a different element.
Types of Radiation • Alpha: attracted toward negatively charged particles • Contain two protons and two neutrons • Have a 2+ charge and a mass of 4 amu • Is equivalent to a helium nucleus • Beta: attracted toward positively charged particles • Are fast moving electrons • Beta decay of Carbon-14 results in Nitrogen and 1 beta particle • Gamma radiation: high energy rays that have no mass and no charge • Accompany alpha and beta particle emission and account for most of the energy lost during radioactive decay.
Nuclear Stability • The ratio of neutrons to protons is a primary indicator of an atom’s stability. • Atoms containing too many or too few neutrons are unstable. • Alpha and beta emissions affect the neutron to proton ratio of a newly created nucleus. • There are few radioactive atoms in nature because most of them have already decayed into stable atoms.