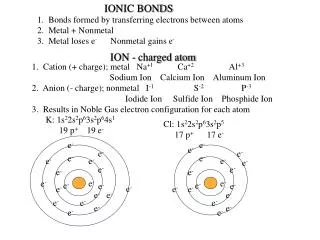
IONIC BONDS
This piece explores the formation of ionic compounds and the specific ratios in which certain elements combine to form stable ions. We examine examples like Lithium (Li) reacting with Fluorine (F) to produce LiF, and Magnesium (Mg) with Fluorine to create MgF2. The investigation prompts questions about the determining factors for these ratios, such as valence electrons. Specifically, we discuss why Li pairs with one F atom while Mg requires two, deepening the understanding of ionic bonding principles and the periodic table's influence on element interactions.

IONIC BONDS
E N D
Presentation Transcript
IONIC BONDS Only certain ratios (combinations) of elements result in the formation of ionic compounds. For ex: Lithium (Li) will react with Fluorine (F) to form LiF. BUT, it will NOT form LiF2, LiF3, etc. Magnesium (Mg) will react with Fluorine to form MgF2. BUT, it will NOT form MgF, MgF3, etc.
IONIC BONDS We are going to investigate what determines the ratios of the elements in Ionic compounds. Q: What do you think determines this ratio? Q: How many valence e- does Li have? How about F? (What is the total # between them?) Q: How many valence e- does Mg have? How about 2 atoms of F? (What is the total # between them?) Q: Why do you think that Li combines with only one F, but Mg needs to combine with 2 F atoms?