Understanding Ionic Bonds: Formulas for Calcium, Aluminum, Strontium, Magnesium, and Potassium
DESCRIPTION
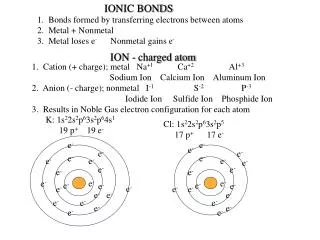
This guide explores the formation of ionic bonds between various elements. We will derive chemical formulas for the following pairs: Calcium (Ca) and Oxygen (O), Aluminum (Al) and Iodine (I), Strontium (Sr) and Silicon (Si), Magnesium (Mg) and Phosphorus (P), and Potassium (K) and Nitrogen (N). Ionic bonds occur when atoms transfer electrons, resulting in the creation of charged ions that attract each other. Understanding these formulas is essential for mastering basic chemistry concepts.
1 / 1
Télécharger la présentation 

Understanding Ionic Bonds: Formulas for Calcium, Aluminum, Strontium, Magnesium, and Potassium
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
IONIC BONDS FOR A GRADE!!! Write chemical formulas for: 1.) Ca and O 2.) Al and I 3.) Sr and Si 4.) Mg and P 5.) K and N
More Related