ENERGY BALANCE MODELS
ENERGY BALANCE MODELS. Balancing Earth’s radiation budget offers a first approximation on modeling its climate Main processes in Energy Balance Models (EBMs) are: Radiation fluxes Equator-to-pole transport of energy .

ENERGY BALANCE MODELS
E N D
Presentation Transcript
Balancing Earth’s radiation budget offers a first approximation on modeling its climate • Main processes in Energy Balance Models (EBMs) are: • Radiation fluxes • Equator-to-pole transport of energy
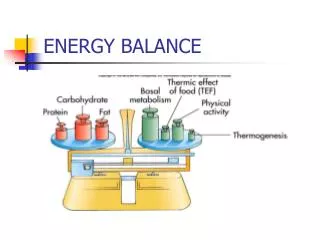
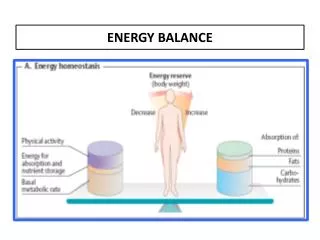
The simplest way is looking at the Earth’s climate in terms of its global energy balance • Over 70 % of the incoming energy is absorbed at the surface surface albedo plays a key role , being the ratio between outgoing and incoming radiation • The output of energy is controlled by • Earth’s temperature • Transparency of the atmosphere to this outgoing thermal radiation
There are two forms of EBM: • Zero-dimensional model The Earth is considered as a single point with a mean effective temperature • First-order model The temperature is latitudinally resolved
Solar radiation input: Si = pR2S • Reflected solar radiation: Sr = a* Si • Emitted infrared radiation: E = 4pR2sTe4 R = distance between Earth and Sun, Te = effective temperature, s Stefan-Botlzman constant, S = solar constant = 1370 W/m2
Therefore, (1-a)*(S/4)=sTe4 Example: DT = 33 K, a = 0.3 Ts = 288 K Note a is the albedo. When describing models we will use a terminology according to McGuffie and Henderson-Sellers
Note that Ts = Te + DT with Te being the effective temperature and DT the greenhouse increment. In other words, the effective temperature (e.g., in a simplistic way the ‘body planet’ temperature) is lower than Ts (the Earth+greenhouse temperature)
Trip to Venus • S = 2619 W/m2 • a = 0.7 • Te = ?
Te = 242 K • Though Venus is closer to the Sun, it has a lower Te than Earth because of the high albedo as it is completely covered by clouds • Besides, Venus atmosphere is very dense and made mostly of carbon dioxide (CO2) • Ts was found to be 730 K ! • The difference between Te and Ts is partially due to greenhouse and partially to adiabatic warming of descending air
Rate of change of temperature mc (DT/Dt)=(R↓-R↑)Ae Where Ae = area of the Earth, c = specific heat capacity of the system, m = mass of the system, R↓ and R↑ are the net incoming and net outgoing radiative fluxes (per unit area)
Swimming pool warming • How long would it take for your swimming pool to warm by 6 K ? • Let us calculate the warming for each day (Dt = 1) • DT is our unknown • Ae = 30 m x 10 m • Depth = 2 m • c = 4200 J/(Kg*K) total heat capacity C = ro*c*V = ro*c*d*Ae=1000*4200*2*30*10=2.52*109J/K with ro = water density • (R↓-R↑) = 20 W/m2 in 24 hours • 2.52*109 = 20 x 30 x 10 x 24 x 60 x 60 DT (1 day) = 0.2K • DT (1 month) = 0.2 x 30 = 6 K
What about the Earth ? Remember : mc (DT/Dt)=(R↓-R↑)Ae R↑ Stefan-Boltzman R↑ esT4ta With taaccounting for the infrared atmospheric transmissivity R↓ = (1-a)*S/4 DT/Dt =((1-a)*S/4 - esT4ta) /C C = fw*ro*c*d*Ae = 1.05*1023 J/K fw = fraction water 0.7, d = 70 m (depth of mixed layer)
One-dimensional EBM (1-a (Ti))*S(Ti)/4= R↑(Ti)+F(Ti)
The term F(Ti) refers to the loss of energy by a latitude zone to its colder neighbor or neighbors • Plus, any ‘storage’ system have been ignored so far since we have been considering time-scale where the net loss or gain of stored energy is small. • Any stored energy would appear as an additional term Q(Ti) on the right side of the previous equation
Parametrization of the climate system • Albedo a(Ti) = 0.6 if Ti < Tc or 0.3 if T > Tc Tc = critical temperature, ranges between -10ºC and 0ºC
Albedo II Another way for parametrizing albedo is a(Ti) =b(phi)-0.009Ti Ti < 283K a(Ti) =b(phi)-0.009x283 Ti ≥ 283K b(phi) is a function of latitude phi
Outgoing radiation R ↑(Ti) = A+BTi with A and B being empirically determined constants designed to account for the greenhouse effect of clouds, water vapour and CO2
Outgoing radiation II • R(Ti) = sTi4 [ 1-mi*tanh(19*Ti6x10-16)] With mi representing atmospheric opacity
Rate of transport of energy F(Ti) = Kt(Ti-Tav) where T is the global average temperature and Kt is an empirical constant
Box Models: another from of EBM • Ocean – atmosphere system with 4 boxes • 1) Atm over Ocean, 2) Atm. Over land, 3) Ocean mixed layer, 4) Deep ocean
The heating rate of the mixed layer is computed assuming a constant depth of the mixed layer in which the temperature difference DT changes in response to the: 1) change in surface thermal forcing DQ, 2) atmospheric feedback, expressed in terms of a climate feedback parameter l, 3) the leakage of energy permitted to the underlying water
The equations describing the rates of heating in the two layers are therefore: • Mixed layer (total capacity Cm) Cm d(DT)/dt = DQ- lDT-DM 2) Deeper waters DT0/ t = K 2 DT0/ z2 With K being the turbulent diffusion coefficient and assumed constant
DM acts as a surface boundary condition to the eq. 2 of the previous slide • If we assume that DT0(0,t)=DT(t) then DM can be computed as: DM = -grwcwK(DT0/ z)z=0 And can be used in the previous Eq. 1. g is a parameter used to average over land and ocean and ranges between 0.72 and 0.75. rw and cw are the density and specific heat capacity of water
Using this approach it is possible to estimate the impacts of increasing atmopsheric CO2. • If DQ is assumed to increase exponentially DQ=b*t*exp(wt) b and w are coefficients to be determined.
The level of complexity can be increased by including, for example, separate systems for the Northern and Southern hemisphere land, ocean mixed layer, ocean intermediate layer and deep oceans.
Pros: • Includes polar sinking ocean water into deep ocean • Seasonally varying mixed layer depth • Seasonal forcing • Cons • Hemispherically averaged cloud fraction • No opportunity to incorporate temperature-surface albedo feedback mechanism (as land is hemispherically averaged)
Readings: McGuffie and Henderson-Sellers Chapter 3, pp 81 - 116