Understanding the Bohr Atom: Quantum Mechanics and Chemical Bonds
This exploration of the Bohr atom delves into the fundamental principles of atomic theory as proposed by renowned theoretical physicists like Niels Bohr and Edwin Schrödinger. It covers essential topics such as absorption lines, the behavior of electrons, and the nature of chemical bonds as explained by Linus Pauling. Additionally, the interaction of strong nuclear forces, protons, neutrons, and the exchange of gluons are also discussed, highlighting the delicate balance of attraction and repulsion that forms the foundation of atomic structure.


Understanding the Bohr Atom: Quantum Mechanics and Chemical Bonds
E N D
Presentation Transcript
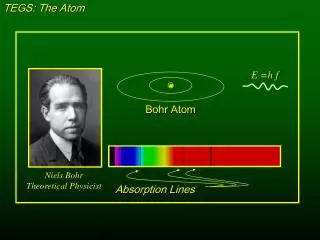
TEGS: The Atom E =h f Bohr Atom Niels BohrTheoretical Physicist Absorption Lines
TEGS: The Atom Quantum Atom
TEGS: The Atom Absorption Lines E =h f Quantum Atom Edwin SchroedingerTheoretical Physicist
TEGS: The Atom - - + + Nucleus “Shared” electrons Linus PaulingTheoretical Chemist Chemical Bond Like charges repel (nuclei, + and +). Unlike charges attract (electron and nucleus, - and +) Attraction = repulsion; balance= Chemical Bond
TEGS: The Atom The Nucleus Strong Nuclear Force Attraction Proton Neutron Electric repulsion
TEGS: The Atom The Nucleus Strong Nuclear Force Attraction Gluon The Strong Nuclear Force occurs because of the exchange of gluons between the protons and neutrons.