Bioinorganic Chemistry
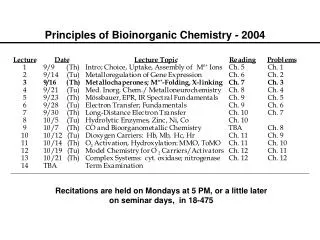
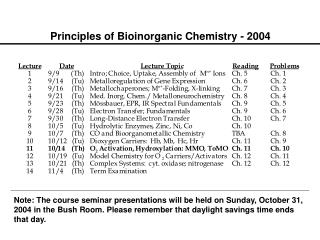
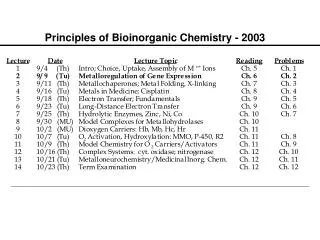
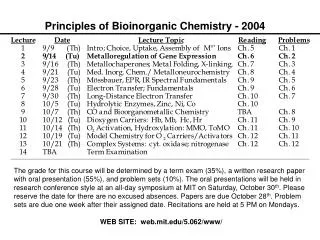
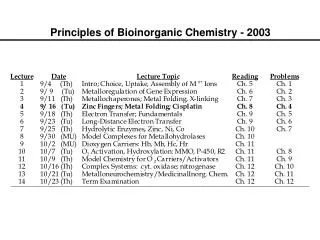
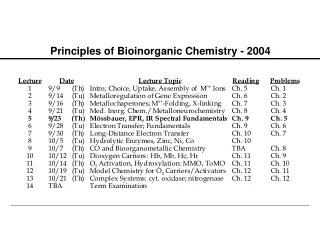
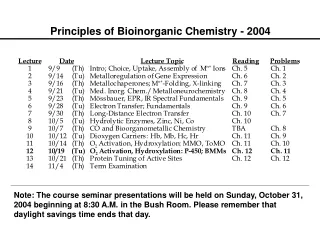
Bioinorganic Chemistry. Chapter 16. Bioinorganic Chemistry. Only a few representative examples will be presented for these bioinorganic compounds and their actions. Table 16-1 (mammalian biochemistry) Examine the importance of iron.

Bioinorganic Chemistry
E N D
Presentation Transcript
Bioinorganic Chemistry Chapter 16
Bioinorganic Chemistry • Only a few representative examples will be presented for these bioinorganic compounds and their actions. • Table 16-1 (mammalian biochemistry) • Examine the importance of iron. • A relatively new area of study for bioinorganic molecules is the incorporation of MO calculations.
Porphyrins and Related Complexes in Bioinorganic Molecules • A porphine ring has a square planar geometry with a “pocket” in the center. • Examine Fig. 16-1. • A metalloporphyrin complex can result by incorporating a metal atom into the pocket (look at heme from Rasmol). • Axial sites are available for other ligands. • Structure, specificity, and reactivity are changed by differing the side chains, metal ions, and surrounding species.
Hemoglobin and Myoglobin • Oxygen transfer and storage agents in the blood and muscle tissue. • Hemoglobin transports oxygen (O2) from the lungs/gills to tissues and muscles. • Myoglobin stores oxygen (O2) in the muscles and tissues. Oxygen commonly transfers from the hemoglobin to the myoglobin for later use.
Hemoglobin • Made up of four globin protein subunits ( and ). • Each protein partially encloses a heme group. • Each heme group is in a porphyrin pocket. • One axial position of the iron is bound to an imidazole nitrogen from the protein. • One axial position is available/vacant or has H2O bound to it. • Dissolved O2 can bind reversibly to this axial position. http://www.umass.edu/microbio/chime/hemoglob/
Hemoglobin • In hemoglobin, the Fe(II) does not become oxidized to Fe(III) or Fe(IV). • Occurs readily in simpler systems (see Figure on the next page). • There needs to be reversible binding of the O2 without oxidation. A free heme also oxidizes in an aqueous environment. • Why doesn’t oxidation occur in hemoglobin by O2 or H2O?
Hemoglobin • In nonoxygenated hemoglobin, the Fe(II) is about 70 pm out of the plane of the porphyrin nitrogens (show with Chime). • Bonding O2 or CO in the sixth position causes the iron to be come planar. • Fe(II) becomes diamagnetic • Oxygen bonds at an angle of ~130 degrees (show with Chime). Explain these structural changes upon bonding.
Hemoglobin • There is a considerable amount of backbonding from the metal to the O2. • Can be described as Fe(III)-O2- • Why is the O2 bent? The energy changes very little with this angle. • suggestions
Hemoglobin • Cooperativity • The function of hemoglobin is to bind O2 at high oxygen pressure and carry it through the blood to needed areas (and myoglobin for storage). Hb + 4O2 Hb(O2)4 Hb(O2)4 + 4Mb 4Mb(O2) + Hb • As one iron binds an oxygen molecule in Hb, the molecular shape changes to make binding of additional oxygen molecules easier. In a similar fashion, initial removal of oxygen triggers the release of the remaining oxygens.
Hemoglobin • At low partial pressures of O2, Mb has a much greater affinity for O2. • The Bohr effect. • Increased acidity favors the release of O2 from Hb(O2)4