Spectra
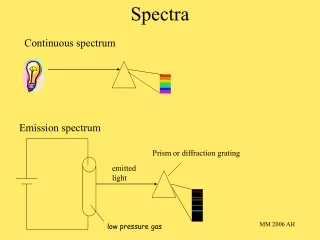
Spectra. White Light. Light from many colors mixes to form white light. Different colors have different wavelengths. Shorter wavelengths bend more through a prism. A diffraction grating combines many edges and acts like a prism. Emitted photon. Moving electron. Sources of Photons.

Spectra
E N D
Presentation Transcript
White Light • Light from many colors mixes to form white light. • Different colors have different wavelengths. • Shorter wavelengths bend more through a prism. • A diffraction grating combines many edges and acts like a prism.
Emitted photon Moving electron Sources of Photons • Accelerated electrons and atoms emit photons. • Photons can be reabsorbed as well.
Temperature • Higher temperature means more energy. • Temperature is measured in kelvin (K). • Absolute zero: 0 K = -273 ºC = -459 ºF. • Room temperature: 300 K 20 ºC = 68 ºF. • Temperature of the Sun: 5800 K 6100 ºC 11,000 ºF • At high temperatures kelvin and centigrade are about the same.
Heated objects give off electromagnetic waves. Higher temperature has more radiation A hot object gives off a spectrum of frequencies. Shifts based on temperature intensity high energy low energy frequency Radiation from Heat
Radiation due a heated object is called blackbody radiation. Wien’s law relaters the temperature to the peak wavelength. intensity high energy low energy frequency Continuous Spectrum
Atoms and Light • An atom is a nucleus surrounded by electrons. • An electron can be in specific energy states. • Change from a high to low energy state produces a photon. • Atoms can also absorb a photon to excite an electron.
Discrete Spectrum • Each atom has its own set of energy levels. • Each atom generates photons at specific frequencies. • The pattern of frequencies (colors) identifies the atom. • Examples include neon or mercury lights. helium neon