Electromagnetic Radiation in Quantum Physics
Explore the properties of electromagnetic radiation, wavelengths, frequencies, and energy levels in quantum mechanics. Learn about photons, quantization of energy, matter waves, and atomic spectra.

Electromagnetic Radiation in Quantum Physics
E N D
Presentation Transcript
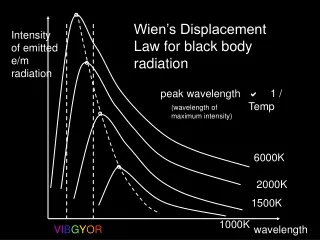
wavelength Visible light Amplitude wavelength Node Ultraviolet radiation Chapter 6: Electromagnetic Radiation
Rank the following in order of increasing frequency: microwaves radiowaves X-rays blue light red light UV light IR light
Waves have a frequency • Use the Greek letter “nu”, , for frequency, and units are “cycles per sec” • All radiation: • = c • c = velocity of light = 3.00 x 108 m/sec • Long wavelength --> small frequency • Short wavelength --> high frequency
What is the wavelength of WONY? What is the wavelength of cell phone radiation? Frequency = 850 MHz What is the wavelength of a microwave oven? Frequency = 2.45 GHz
Quantization of Energy Light acts as if it consists of particles called PHOTONS,with discrete energy. Energy of radiation is proportional to frequency E = h • h = Planck’s constant = 6.6262 x 10-34 J•s
E = h • Relationships:
Rank the following in order of increasing photon energy: microwaves radiowaves X-rays blue light red light UV light IR light
E = h • n What is the energy of a WONY photon?
Energy of Radiation What is the frequency of UV light with a wavelength of 230 nm? What is the energy of 1 photon of UV light with wavelength = 230 nm?
What is the energy of a mole of 230 nm photons? Can this light break C-C bonds with an energy of 346 kJ/mol?
Where does light come from? • Excited solids emit a continuous spectrum of light • Excited gas-phase atoms emit only specific wavelengths of light (“lines”)
The Bohr Model of Hydrogen Atom • Light absorbed or emitted is from electrons moving between energy levels • Only certain energies are observed • Therefore, only certain energy levels exist • This is the Quanitization of energy levels
Emission spectra of gaseous atoms • Excited atoms emit light of only certain wavelengths • The wavelengths of emitted light depend on the element.
Constant = 2.18 x 10-18 J For H, the energy levels correspond to: Energy level diagram:
Each line corresponds to a transition: Example: n=3 n = 2
Explanation of line spectra Balmer series
Matter Waves • All matter acts as particles and as waves. • Macroscopic objects have tiny waves- not observed. • For electrons in atoms, wave properties are important. • deBroglie Equation:
Matter waves Macroscopic object: 200 g rock travelling at 20 m/s has a wavelength: Electron inside an atom, moving at 40% of the speed of light:
Heisenberg Uncertainty Principle • Can’t know both the exact location and energy of a particle • So, for electrons, we DO know the energy well, so we don’t know the location well
Schrodinger’s Model of H • Electrons act as standing waves • Certain wave functions are “allowed” • Wave behavior is described by wave functions: • 2describes the probability of finding the electron in a certain spot • Also described as electron density
Example Wavefunction Equation slightly simplified:
It’s all about orbitals • Each wavefunction describes a shape the electron can take, called an ORBITAL • Allowed orbitals are organized by shells and subshells • Shells define size and energy (n = 1, 2, 3, …) • Subshells define shape (s, p, d, f, …) • Number of orbitals is different for each subshell: s = 1 orbital p = 3 orbitals d = 5 orbitals f = 7 orbitals
Which subshell does not exist? • 5s • 2p • 2d • 4f • 15s
NODES Spherical Nodes