Inosilicates: single chains- pyroxenes
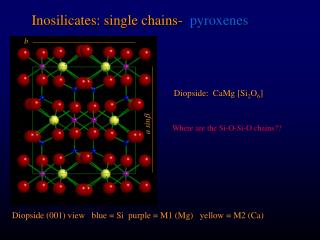
b. Diopside: CaMg [Si 2 O 6 ]. a sin . Where are the Si-O-Si-O chains??. Inosilicates: single chains- pyroxenes. Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca). b. a sin . Inosilicates: single chains- pyroxenes.

Inosilicates: single chains- pyroxenes
E N D
Presentation Transcript
b Diopside: CaMg [Si2O6] a sin Where are the Si-O-Si-O chains?? Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
b a sin Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
b a sin Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
b a sin Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
b a sin Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
b a sin Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
Perspective view Inosilicates: single chains- pyroxenes Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
SiO4 as polygons (and larger area) IV slab VI slab IV slab a sin VI slab Inosilicates: single chains- pyroxenes IV slab VI slab IV slab b Diopside (001) view blue = Si purple = M1 (Mg) yellow = M2 (Ca)
M1 octahedron Inosilicates: single chains- pyroxenes
M1 octahedron Inosilicates: single chains- pyroxenes
(+) M1 octahedron (+) type by convention Inosilicates: single chains- pyroxenes
M1 octahedron This is a (-) type (-) Inosilicates: single chains- pyroxenes
T M1 T Creates an “I-beam” like unit in the structure. Inosilicates: single chains- pyroxenes
(+) T M1 T Creates an “I-beam” like unit in the structure Inosilicates: single chains- pyroxenes
(+) (+) (+) (+) (+) Inosilicates: single chains- pyroxenes The pyroxene structure is then composed of alternating I-beams Clinopyroxenes have all I-beams oriented the same: all are (+) in this orientation Note that M1 sites are smaller than M2 sites, since they are at the apices of the tetrahedral chains
(+) (+) (+) (+) (+) Inosilicates: single chains- pyroxenes The pyroxene structure is then composed of alternation I-beams Clinopyroxenes have all I-beams oriented the same: all are (+) in this orientation
Inosilicates: single chains- pyroxenes Tetrehedra and M1 octahedra share tetrahedral apical oxygen atoms
Inosilicates: single chains- pyroxenes The tetrahedral chain above the M1s is thus offset from that below The M2 slabs have a similar effect The result is a monoclinic unit cell, hence clinopyroxenes (+) M2 c a (+) M1 (+) M2
Inosilicates: single chains- pyroxenes Orthopyroxenes have alternating (+) and (-) I-beams the offsets thus compensate and result in an orthorhombic unit cell This also explains the double a cell dimension and why orthopyroxenes have {210} cleavages instead of {110) as in clinopyroxenes (although both are at 90o) c (-) M1 (+) M2 a (+) M1 (-) M2
Clinpyroxene vs. orthopyroxene structures C2/c e.g. diopside CaMgSi2O6 Pbca e.g. enstatite Mg2Si2O6
Alternative clinopyroxene structure (P21/c) The C2/c and P21/c structures differ in the way the tetrahedral chains are kinked. In the C2/c structure the chains are relatively straight and are all symmetry-related to each other. In the P21/c structure, chains in the same (100) layer are kinked in opposite senses, so they are no longer symmetry-related. 174° 149° 170°
The orthopyroxene solid solution : Enstatite (MgSiO3) to ferrosilite (FeSiO3)
M1 M2 Low temperature Mg Fe2+ Intermediate temperature Mg1-xFe2+x MgxFe2+1-x “Infinite” temperature Mg0.5Fe2+0.5 Mg0.5Fe2+0.5 Non-convergent cation ordering in orthopyroxenes Fe2+ is slightly larger than Mg and prefers to sit on the larger M2 site (i.e. the crystal has a lower enthalpy when Fe2+ is sitting on M2). Example: For a composition (Mg0.5Fe2+0.5)SiO3 We can measure “x” experimentally and use it to determine what the cooling rate and effective equilibration rate of the mineral was (geospeedometry).
Pigeonite and augite solid solutions Hypersthene transforms to the pigeonite (C2/c) structure at high temperatures. Pigeonite has an expanded M2 site, and can accept larger amounts of Ca substituting for (Mg, Fe2+). The endmembers diopside (CaMgSi2O6) and hedenbergite (CaFeSi2O6) are both clinopyroxenes (C2/c). Ca occupies M2 and there is complete solid solution between Mg and Fe2+ on M1. The term augite is used to describe the Ca-rich clinopyroxene solid solution. Pigeonite and augite are separated by a large miscibility gap because of the large difference between the radius of (Mg, Fe) and Ca
Phase diagram for the pigeonite-diopside “binary” At high temperature, both pigeonite and augite are monoclinic with the same C2/c structure. Miscibility between these two endmembers is limited due to the large difference in the ionic radii of (Mg, Fe) and Ca. The eutectic melting loops are typical features of the solidification of a solid solution with limited miscibility. Mg 0.86 Å 32% Ca 1.14 Å
Reconstructive phase transition and low-T eutectoid point In low-Ca pigeonite, the M2 site is too large for the small Mg and Fe cations. The mismatch is tolerated at high temperature because thermal vibration of the Mg and Fe atoms prevents the structure from collapsing. At low temperature there is a reconstructive phase transition to the orthopyroxene (hypersthene structure, which has a much smaller M2 site. The reconstructuve phase transition leads to the development of a eutectoid point.
Crystallographic and optical axes align C crystallographic axis at 32 to 42º angle to the Z optical axis Pigeonite – CPX - Monoclinic OPX - Orthorhombic
Reconstructive phase transition and low-T eutectoid point The reconstructuve phase transition from monoclinic pigeonite to orthorhombic hypersthene is very slow, and will only occur in very slowly-cooled rocks. If the transition doesn’t take place, the structure needs another way of coping with the small Mg and Fe cations in M2. Displacive phase transition to the low pigeonite structure (P21/c) occurs instead. Transition temperature decreases with increasing Ca content, as the larger Ca atoms hold the structure apart.
Exsolution phenomena in pyroxenes (Mg, Fe)-Ca diffusion can occur, therefore augite exsolution lamellae develop on entering the miscibility gap. Lamellae are parallel to (001) of monoclinic host. No time for reconstructive phase transition. Displacive phase transition occurs below Tc in the pigeonite component of the intergrowth (Mg, Fe)-Ca diffusion can occur. Augite lamellae develop parallel to (001) of monoclinic host. Reconstructive phase transition occurs in pigeonite component of the intergrowth. Further exsolution of augite occurs // (100) of orthorhombic host. No time for (Mg, Fe)-Ca diffusion, therefore no exsolution. No time for reconstructive phase transition. Displacive phase transition from high to low pigeonite occurs below Tc
Microstructures of exsolved pyroxenes The exsolving phase forms as lamellae (thin slabs). The orientation of the lamellae is determined by the plane of best fit between the two phases. Augite in pigeonite (and vice versa) has best fit close to (001) Augite in hypersthene (and vice versa) has best fit close to (100)
Coherence at interfaces • Coherent/semi-coherent/incoherent interfaces: these terms are based on the degree of atomic matching across the interface. • Coherent interface means an interface in which the atoms match up on a 1-to-1 basis (even if some elastic strain is present). • Incoherent interface means an interface in which the atomic structure is disordered. • Semi-coherent interface means an interface in which the atoms match up, but only on a local basis, with defects (dislocations) in between.
Coherent interface Incoherent interface Semi-coherent interface
Homophase vs. Heterophase • There is a useful comparison that can be made between grain boundaries (homophase) and interphase boundaries (heterophase).Structure Grain Boundary Interface atoms no boundary coherentmatch (or, S3 coherent twin in fcc) interface dislocations low angle g.b. semi- coherent disordered high angle g.b. incoherent • Remember: for a grain boundary to exist, there must be a difference in the lattice position (rotationally) between the two grains. An interface can exist even when the lattices are the same structure and in the same (rotational) position because of the chemical difference.
LAGB to HAGB Transition • LAGB: steep risewith angle.HAGB: plateau Disordered Structure Dislocation Structure
b Read-Shockley model • Start with a symmetric tilt boundary composed of a wall of infinitely straight, parallel edge dislocations (e.g. based on a 100, 111 or 110 rotation axis with the planes symmetrically disposed). • Dislocation density (L-1) given by:1/D = 2sin(q/2)/b q/b for small angles. D Phase Transformation
Read-Shockley, contd. • For an infinite array of edge dislocations the long-range stress field depends on the spacing. Therefore given the dislocation density and the core energy of the dislocations, the energy of the wall (boundary) is estimated (r0 sets the core energy of the dislocation):ggb = E0 q(A0 - lnq), whereE0 = µb/4π(1-n); A0 = 1 + ln(b/2πr0) Phase Transformation
High angle g.b. structure • High angle boundaries have a disordered structure. • Bubble rafts provide a useful example. • Disordered structure results in a high energy. Low angle boundarywith dislocation structure Phase Transformation
Mechanism of eutectoidal decompostion of pigeonite to augite and orthopyroxene
Microstructures of exsolved pyroxenes Exact orientation depends on lattice parameters of the two phases. The orientation varies systmatically with temperature. This can be used to constrain the temperature at which a particular generation of lamellae grew.
Microstructures of exsolved pyroxenes The thickness and spacing of lamellae depends on temperature and the time available for them to grow. By performing annealing experiments and measuring the wavelength of exsolution features using transmission electron microscopy, we are able to calibrate the changes in wavelength as a function of isothermal annealing time. For a process determined by volume diffusion, we observe that the spacing of lamellae is proportional to (time)1/3.
Example: Cooling rate of chondrules in the Allende carbonaceous chondrite Chondrules are a major component of chondritic meteorites. It is believed that chondrules formed in the solar nebula prior to accretion of the meteorite parent bodies. Chondrules are thought to have formed by crystallisation of melt droplets. Pyroxene textures from granular olivine-pyroxene chondrules (GOP’s) provide constraints on their cooling history and therefore provide information about the conditions in the solar nebula. Wavelengths between 25 and 33 nm are observed, translating to cooling rates between 25 and 0.4 °C/hour over the temperature range 1350-1200 °C. No orthopyroxene suggests more rapid cooling (>104 °C/hour) below 1000 °C.
Example: Cooling rate of chondrules in the Allende carbonaceous chondrite
Microstructures of exsolved pyroxenes Inverted Pigeonite
Pyroxene Chemistry Jadeite Aegirine NaAlSi2O6 “Non-quad” pyroxenes NaFe3+Si2O6 0.8 Omphacite aegirine- augite Spodumene: LiAlSi2O6 Ca / (Ca + Na) Ca-Tschermack’s molecule 0.2 CaAl2SiO6 Augite Diopside-Hedenbergite Ca(Mg,Fe)Si2O6