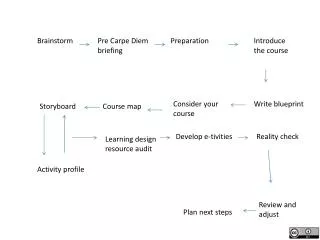
Brainstorm
Brainstorm. What is chemistry? What will you see this unit? What will you hear discussed? What will you do this unit?. Topic 1.0 Lab Safety and Properties. Outcomes. 2.1k- What are caustic materials? What are some reactions that should be avoided?


Brainstorm
E N D
Presentation Transcript
Brainstorm What is chemistry? What will you see this unit? What will you hear discussed? What will you do this unit?
Outcomes • 2.1k- What are caustic materials? What are some reactions that should be avoided? • 2.4s- Demonstrate knowledge of WHMIS standards, by using proper techniques for handling and disposing of laboratory materials • AF. Safety- Show concern for safety in planning, carrying out and reviewing activities
1.1 Identification of Safety Issues • 1) Independently create a list of the safety hazards that appear on the next slide. • 2) When asked, turn to your partner and share your ideas • 3) Share as a whole class.
Lab Safety – Mind Map • Create a mind map by adding lab safety ideas to the following topics
Lab Rules Using the mind maps you created, come up with 5 rules for working safely in the lab Share as a class
Lab Safety Video • http://youtu.be/xJG0ir9nDtc
Safety Scenarios • With your assigned group, choose one scenario • Create a skit to explain your scenario and the right and wrong way to handle the situation • Be ready to share!
Safety Rules • 1. Read & know procedure. • 2. Wash hands after lab. • 3. No food or drink in the lab – no tasting! • 4. Wear safety googles always. • 5. Waft substances to smell them. • 6. Tie back long hair. • 7. No flip-flops or open shoes • 8. Place test tubes in a test tube rack when transporting and pouring in liquid.
Safety Rules • 9. Wipe up spills. • 10. Wipe off counter after lab. • 11. Place chipped/broken glass in broken glass pail. • 12. Label ALL beakers/test tubes. • 13. Report all accidents/spills. • 14. Stay at your lab bench with your group. • 15. Never leave lab bench unattended.
Household Symbols • These symbols are on household products: Shapes: Caution Danger Warning
WHMIS Symbols • Workplace Hazardous Materials Information System • Differs from household symbols – • WHMIS is worldwide! • MSDS- Material Safety Data Sheets • Information about each chemical used • Ex. Flammability, volatility, mp, bp
WHMIS symbols Where would you see each of these symbols?
Special Note: • Caustic – ability to burn or corrode tissue ; refers to strong bases NaOH • Corrosive – ability to corrode; mainly refers to acids HCl Corrode – means to “gnaw at” (Latin)
Reactions to AVOID • Bleach X ammonia = Chlorine gas (BAD) • Used as a chemical warfare agent in WWII – corrosive especially when inhaled X +
Assignment • Pg. 96 qs. 3, 4, 5, 6, 7 • Review as a class when finished
1.2 Classifying Matter • 1.2k -How can we classify materials based on properties? • 1.2k - How can we classify materials based on their composition?
Try It – On a spare piece of paper How would you group/classify the following items? *Make sure to jot down your justification for your groups.*
1.2 Classifying Matter • Matter – any substance that occupies space and has mass • Ex. Air, Water • Example of non-matter is: Skills Ideas Beliefs Energy
1.2 Classifying Matter Particle Model of Matter (review) • Matter is made of particles • Particles attract each other • Particles are always moving • Particles move faster with higher temperatures • Each pure substance has different particles than another substance
1.2 Classifying Matter A. States of Matter Sublimation Melting Evaporation Solid Liquid Gas Freezing Condensation Deposition
1.2 Classifying Matter • Draw the particles within the beaker to demonstrate the particle model of matter and states of matter Solid Gas Liquid
1.2 Classifying Matter B. Composition Made of 1 kind of matter Combo of pure substances Particles are suspended & hard to separate One kind of atom Particles suspended but can be separated Two or more atoms/elements in a fixed proportion Looks like one substance Different parts are visible
1.2 Classifying Matter Examples Oxygen O2 Grape jelly Italian salad dressing H20 Vinegar Choc chip cookie
1.2 Classifying Matter • Solutions can be in different states – don’t let this fool you!
1.2 Classifying Matter C. Properties of Matter • Matter can be identified and classified based on physical and chemical properties
LAB: Identifying Mystery Substance • Turn to pg. 100-101 • Pre-lab: • Title • Question • Outcome – look on outcome sheet and write out the outcome the lab is building on • Read through procedure – be ready to answer questions before you are able to enter the lab • Safety – list one safety concern of lab and PPE suggested
LAB: Identifying Mystery Substance • After the Lab: • Analysis: Answer qs. 20, 21, 23 • Make sure these are in complete sentences and you are justifying your answers.
Assignment • Pg. 104 • Questions: 3. 4. 6. 7. 8. 9. 10
1.3 Changes in Matter Outcomes • 1.3k- What evidence shows that properties of a material are changed? • 1.3k - How do you know if a new substance has been made? Try IT: Turn to pg. 105 Skill Practice
Practice: Label each of the following as either a Physical (P) or Chemical ( C) change
LAB: Investigating Physical and Chemical Changes • Read through pgs. 106-107 • Pre-lab: • Title • Question • Objective – check outcome sheet • Evidence Table ** changes: Omit: Changes during and after – make into one column called “Evidence”
LAB: Investigating Physical and Chemical Changes • After: • Write in evidence table whether physical or chemical • In ANALYSIS, provide reasoning for each reaction
Assignment • Pg. 109 qs. 2, 4