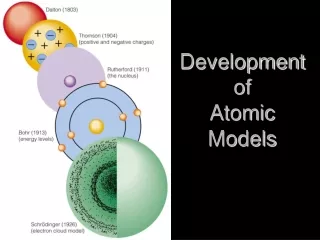
Development of Atomic Models
This overview chronicles the evolution of atomic theory, beginning with Democritus and his concept of the "atomos," meaning indivisible matter, around 400 BC. It covers John Dalton's atomic theory in the early 1800s, which introduced the idea that matter is composed of indivisible atoms. The description of the atom continued to evolve, thanks to JJ Thomson's discovery of the electron and the Plum Pudding Model, followed by Ernest Rutherford's Gold Foil Experiment leading to the Nuclear Model. Finally, Niels Bohr introduced the Planetary Model, detailing electron energy levels and the significance of spectral lines.

Development of Atomic Models
E N D
Presentation Transcript
Democritus • Greek philosopher • 400 BC • “Atomos” concept
Can matter can be divided forever? • Eventually, a piece would be “indivisible” • “Atomos,” meaning “not to be cut,” is smallest piece of matter
John Dalton (early 1800’s) • Coined the term “atom”.
Dalton’s Atomic Theory • Matter made of tiny indivisible particles called “atoms”. • Atoms of one element are alike, and different from atoms of other elements.
Compounds form when different atoms combine in fixed proportions. • Chemical reactions involverearrangement of atoms. • Atoms can’t be created or destroyed, but are conservedin chemical reactions.
Dalton’s Atomic Theory called “Hard Spheres Model”
Thomson’ Experiments • Studied “cathode rays” (electric current) in a “Crooke’s Tube”. • Fluorescent screen, shows how cathode ray behaved in a magnetic field. Lets draw a typical Crooke’s Tube in our notes.
Cathode Rays were negatively charged Cathode Ray Tube and Magnet http://youtu.be/XU8nMKkzbT8 They bent toward (+) plate
Cathode Rays were particles They couldn’t pass through matter.
JJ is Awesome • Concluded the negative “cathode ray” particles came from within atoms. • Discovered first subatomic particle (electron).
What about the Positive? • But…matter is neutral. • Therefore: • A positive charge must exist to balance the negative.
Plum Pudding Model Atoms are positively charged spheres with negatively charged particles scattered throughout.
Yummy… Brian Cox: Thompson and Discovery of Electron http://youtu.be/IdTxGJjA4Jw
Ernest Rutherford (1908) • Physicist who worked in new field of radioactivity.
Found 3 Different Types of Radiation • Used magnetic field to isolate three types of radiation. • Alpha (α) • Beta (β) • Gamma (γ)
Charges of Radiation • The radiation had different charges. Identify the charge each type of radiation has.
Gold Foil Experiment • Shot alpha particles, at very thin piece of gold foil. • Alpha particles have a positive charge, and a mass of 4 amu • Fluorescent screen shows where the particles went. Rutherford Gold Foil http://www.youtube.com/watch?v=5pZj0u_XMbc
Observation: Most alpha particles passed straight through gold foil. Conclusion: Atom’s volume is mostly empty space.
Observation: A few alpha particles deflected at an angle or bounced back. Conclusion: Atoms have a very small, dense positively charged nucleus. http://www.kentchemistry.com/moviesfiles/Units/AtomicStructure/Rutherford3.htm
Nucleus is extremely small compared to the size of the atom as a whole. Deflections happened rarely (1/8000). Modern Example of Gold Foil Experiment in Action http://youtu.be/XBqHkraf8iE
The Nuclear Model Rutherford’s Model is called the “Nuclear Model” Brian Cox: Rutherford and the Nucleus http://youtu.be/wzALbzTdnc8
Comparison to Thomson • Positively charge only contained in nucleus. • Negatively particles scattered outside nucleus. • Charge is not disbursed evenly.
Niels Bohr (1913) • Came up with the “Planetary Model”
Bohr’s Theory • Electrons circle nucleus in specific energy levels or “shells”. • The higher the “energy level,” the higher the electron’s energy.
Energy Levels • Different energy levels can contain different numbers of electrons.
How many per level? • n = the number of the energy level 2n2 = maximum number of electrons an energy level can hold. Ex: Level 3 can hold 2(3)2 = 18 electrons
Draw a Bohr Atom • Ex: The Fluorine Atom (F) • Protons = 9 • Neutrons = 10 • Electrons = 9 • How many energy levels do you draw? • How many electrons in each level? Human Bohr Model http://www.youtube.com/watch?v=PLpZfJ4rGts
Draw a Bohr Ion • They only difference is that one or more electrons gets added or taken out of the outer energy level. • Ex: The Magnesium Ion (Mg+2) • Protons = 12 • Neutrons = 12 • Electrons = 10
(+) Ions (cations) (+) ions are smaller Lost electron(s)
(-) Ions (anions) (-) ions are larger Gained electron(s)
How Did Bohr Come Up With His Model? • Studied the spectral lines emitted by various elements (especially Hydrogen)
What are Spectral Lines? • Energy gets absorbed by an atom causing it to emit a unique set of colored lines. • Used to identify what elements are present in a sample. (elemental “Fingerprint”)
What Causes Spectral Lines? Jumping Electrons!! Video of Line Spectra of Hydrogen http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/linesp16.swf
Jumping Electrons Electrons normally exist in the lowest energy level possible called the “ground state”. (stable) “Ground state” e- configurations are written on the periodic table for each element. Ex: Aluminum is 2-8-3 Calcium is 2-8-8-2
An Electron Gets “Excited” Electrons can absorb a photon (or “quanta”) of energy and “jump up” to a higher energy level farther from the nucleus. This is called the “excited state”. (unstable)
Jumping Electrons • They quickly “fall back down” to the ground state. (stable) • They emit a photon (or “quanta”) of energy that corresponds to how far they jumped. Spectral Lines http://www.youtube.com/watch?v=QI50GBUJ48s
This photon of energy is seen as a spectral line! • Each spectral line corresponds to a specific photon of energy that is released. Model Of Hydrogen Atom and Electrons Jumping http://www.upscale.utoronto.ca/PVB/Harrison/BohrModel/Flash/BohrModel.html
REMEMBER Absorb Energy Jump Up Emit Energy Fall Down
Excited vs Ground State • Periodic table lists ground state electron configurations for neutral atoms. • To recognize an “excited state” configuration, count the electrons and see if the configuration matches the one on the table. • Ex: 2-8-7-3 = 20 electrons • Calcium (atomic # 20) is 2-8-8-2 • So this must be showing one of the ways calcium could be in the excited state.
Valence Electrons • Electrons in highest occupied energy level. • Involved in forming bonds with other atoms. • Atoms are most stable when they obtain a “stable octet” of 8 valence electrons • Noble Gases: (Group 18) • Have stable octet already and are “inert” and unreactive • Ex: Argon 2-8-8, Neon 2-8
Valence Electrons • Look at the last number in the atom’s electron configuration to determine the number of valence electrons. • Ex: • Al 2-8-3 3 valence • Ca 2-8-8-2 2 valence • F 2-7 7 valence
Lewis Dot Diagrams • Shows the number of valence electrons an atom has as “dots” around the atom’s symbol. Phosphorus is 2-8-5
Kernel • Nucleus and non-valence electrons • Inner part of atom not involved directly in reactions • Ex: • Al 2-8-3 has 10 kernel electrons and 3 valence electrons
The Nature of Light • Study of light has provided important information about the structure of atoms. • Dual Nature of Light: • behaves as both waves and as particles (depending on what type of experiment is being performed.) • Speed of Light: all light waves travel at the same velocity • C = 3.0 x 108 meters/sec
Electromagnetic Spectrum • Spectral lines can come from all areas of the EM Spectrum. • Lines of visible colors make up only a small part of the spectrum.