Understanding Alpha Decay: Mechanisms, Energies, and Calculations
This document elucidates the process of alpha decay in heavy nuclei, detailing the conservation of energy during decay and providing calculations for kinetic energy of emitted alpha particles. Using examples such as plutonium-236, we explore the Q-value for various decay modes, analyze potential energy barriers, and introduce quantum mechanics concepts related to tunneling probabilities for alpha emission. Additionally, we offer insights into decay energies and half-lives for various isotopes, enhancing the understanding of nuclear stability and decay processes in heavy elements.

Understanding Alpha Decay: Mechanisms, Energies, and Calculations
E N D
Presentation Transcript
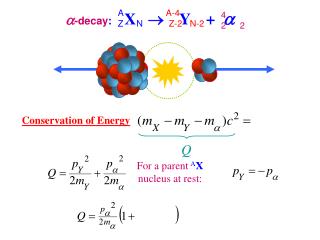
-decay: X Y + A Z A-4 4 2 N Z-2 N-2 2 Conservation of Energy Q For a parent AX nucleus at rest:
Note: for heavy nuclei to within~98%accuracy, anyway We’ll see from a few examples that typically T 4-5 MeV
Repeating anOLDIE but GOODIEfrom Lecture 13 on “Radiation”: 236 94 Is Puunstable to -decay? 236 94 232 92 4 2 Pu U + + Q Q = (MPu – MU-M)c2 = (236.046071u – 232.037168u – 4.002603u)931.5MeV/u = 5.87 MeV > 0
Some (especially the heaviest) nuclei are unstable with respect to the emission of heavy particles • essentially the break up of a nucleus. In one extreme: the emission of a single nucleon but it includes the far more commonalpha emission and fission of the original nucleus into smaller, approximately equal sized nuclei.
Table 8.1 Energy Release (Q value) for various modes of decay of232U Emitted particle Energy Released (MeV) n -7.26 MeV 1H -6.12 2H -10.70 3H -10.24 3He -9.92 4He +5.51 MeV 5He -2.59 6He -6.19 6Li -3.79 7Li -1.94
repulsive Coulomb potential attractive nuclear potential
Let’s follow: The calculation of the kinetic energy of an alpha particle emitted by the nucleus 238U. The model for this calculation is illustrated on the potential energy diagram at the right. stepping through the details:
This potential energy curve combines a nuclear well of radius 7.75 fm (from R = 1.25 x A1/3 fm) and the Coulomb potential energy of an alpha in the electric field of the daughter 234Th nucleus. The mean binding energy per nucleon B/A for 238U (from the Semi-empirical mass formula) is 7.5 MeV. Thus to remove 4 average nucleons would require 30 MeV.
Compare to using the semi-empirical mass formula to calculate the energy required to remove 2 protons and 2 neutrons from the highest238U energy levels. -24.4 MeV assumes they are the last two particles of each type added to the234Thnucleus.
N N N N For the alpha particle Dm= 0.03035 u which gives 28.3 MeV binding energy! Alpha particle protons 2 1.00728 u neutrons 2 1.00866 u Mass of parts 4.03188 u Mass of alpha 4.00153 u 1 u = 1.66054 10-27 kg = 931.494 MeV/c2 The model for alpha emission proposes that the alpha particle is preformed inside the nucleus.
The binding energy released (28.3 MeV) appears in part as kinetic energy of the alpha. An alpha particle with positive energy is created inside the nucleus where it is trapped by the potential barrier. According to quantum mechanics it has a finite probability of escape. Let’s see how well quantum mechanics and our model of the potential can calculate that probability (decay rate)
Tunneling finite (but small) probability of being found outside the nucleus at any time always some probability of a piece of the nucleus escaping the nuclear potential Nuclear potential Coulomb potential with a STATIC POTENTIAL this probability is CONSTANT!
Let’s examine this through a simple model: an forms inside the nucleus and then escapes through quantum mechanical barrier penetration. The potential seen by the is spherically symmetric, so we can start by first separating the variables - the functions Ylm are the same spherical harmonics you saw for the wavefunctions for the hydrogen atom. Then the equation for the radial function Rnl(r) can be written as
For states without orbital angular momentum ( l = 0) this reduces to an equation like that for a 1-dimensional barrier. The transmitted part of the wave function X is of the form where The integral is carried out over the range of the potential barrier. A solution can be found by approximating the shape of the potential as a succession of thin rectangular barriers. In this case the inner limit of the integral is effectively the nuclear radius R, and the outer limit is taken as the point at which the ’s kinetic energy is equal in magnitude to its potential energy.
In simple 1-dimensional case V E III II I where
In simple 1-dimensional case V E x = r1 x = r2 III II I probability of tunneling to here
the point at which the ’s kinetic energy is equal to its potential energy. Where E r2 R So let’s just write as
hence with E=Ta becomes then with the substitutions:
Performing the integral yields: and for R << r2 the term in the square brackets reduces to
When the result is substituted into the exponential the expression for the transmission becomes
The decay probability is = f X where f is the frequency with which the alpha particle hits the inside of the barrier. Thus fcan be estimated from crude time between striking nuclear barrier of 4-8 MeV “pre-formed” alpha Easily giving estimates for l = 106/sec – 10-21/sec
Some Alpha Decay Energies and Half-lives Isotope Ta(MeV) t1/2l(sec-1) 232Th 4.01 1.41010 y 1.610-18 238U 4.19 4.5109 y 4.910-18 230Th 4.69 8.0104y 2.810-13 238Pu 5.50 88years 2.510-10 230U 5.89 20.8 days 3.910-7 220Rn 6.29 56 seconds 1.210-2 222Ac 7.01 5 seconds 0.14 216Rn 8.05 45.0msec 1.5104 212Po 8.78 0.30msec 2.3106 216Rn 8.78 0.10msec 6.9106
this quantum mechanically-motivated relation should be compared with the emperical Geiger-Nuttall law
232Th Q=4.08 MeV =1.4×1010 yr 218Th Q=9.85 MeV =1.0×10-7 sec The dependence of alpha-decay half-life on the kinetic energy of the alpha particle. Values are marked for some isotopes of thorium.
For each series of isotopes the experimental data agree (1911)
The potential seen by an electron in the hydrogen atom is spherically symmetric (depends only on r, not its direction)! Recognizing that we write Schrödinger’s equation in spherical polar coordinates To solve we apply a separation of variables:
=K (some constant) =K2 (also some constant)
Then the problem becomes finding solutions to the separate “stand alone” equations each of which uniquely constraints the wavefunction: A solution to the radial equation can exist only when a constant arising in its solution is restricted to integer values (giving the principal quantum number) Similarly, a constant arises in the colatitude equation giving an orbital quantum number Finally, constraints on the azimuthal equation give a magnetic quantum number
Let’s examine this through a simple model: an forms inside the nucleus and then escapes through quantum mechanical barrier penetration. The potential seen by the is spherically symmetric, so we can start by first separating the variables - the functions Ylm are the same spherical harmonics you saw for the wavefunctions for the hydrogen atom. Then the equation for the radial function Rnl(r) can be written as
ALL are equivalent to a reflection (axis inversion) plus a rotation PARITY TRANSFORMATIONS y y x' x x z z y y x x z z y' y y z' x x' x The PARITY OPERATOR on 3-dim space vectors every point is carried through the origin to the diametrically opposite location z z y'
Wave functions MAY or MAY NOT have a well-defined parity (even or odd functions…or NEITHER) P P = +1 P P = -1 but the more general P
However for any spherically symmetric potential, the Hamiltonian: H(-r) = H(r) H(r) → → [ P, H ] = 0 So they bound states of such a system have DEFINITE PARITY! That means, for example, all the wave functions of the hydrogen atom!
-dE/dx = (4pNoz2e4/mev2)(Z/A)[ln{2mev2/I(1-b2)}-b2] I = mean excitation (ionization) potential of atoms in target ~ Z10 GeV 10 8 6 4 3 2 1 Minimum Ionizing: -dE/dx [ MeV·g-1cm2] MeV g/cm2 1 – 1.5 0.01 0.1 1.0 10 100 1000 Muon momentum [ GeV/c ]
A typical gamma detector has a light-sensitive photomultiplier attached to a small NaI crystal. The scinitillator responds to the dE/dx of each MIP track passing through
If an incoming particle initiates a shower, each track segment (averaging an interaction length) will leave behind an ionization trail with about the same energy deposition. The total signal strength Number of track segments Basically Measuring energy in a calorimeter is a counting experiment governed by the statistical fluctuations expected in counting random events.
Since E Ntracks andN = N we should expectE E E E 1 E E E and the relative error = E = AE a constant that characterizes the resolution of a calorimeter
Y(r)=c(r)ml(q,f )the angular part of the solutions are theSPHERICAL HARMONICS ml(q,f ) = Pml (cosq)eimf Pml (cosq) = (-1)msinmq[()mPl (cosq)] (2l + 1)( l- m)! 4p( l + m)! d d (cosq) 1 2l l! d d (cosq) Pl(cosq) = [()l(-sin2q)l ]
The Spherical Harmonics Yℓ,m(,) ℓ = 3 ℓ = 0 ℓ = 1 ℓ = 2
z then note r -r means q p - q f p + f q p-q y f so: eimf eimfeimp x (eip)m=(-1)m and: Pml (cosq) Pml (cos(p-q)) = Pml (-cosq) (-sin2q)l = (-1-cos2q)l butd/d(cosq)-d/d(cosq)
(2l + 1)( l- m)! 4p( l + m)! ml(q,f ) = Pml (cosq)eimf Pml (cosq) = (-1)m(1-cos2q)m[()mPl (cosq)] d d (cosq) 1 2l l! d d (cosq) Pl(cosq) = [()l(-sin2q)l ] So under the parity transformation: P:ml(q,f ) =ml(p-q,p+f)=(-1)l(-1)m(-1)m ml(q,f ) = (-1)l(-1)2m ml(q,f ) )=(-1)l ml(q,f ) An atomic state’s parity is determined by its angular momentum l=0 (s-state) constant parity = +1 l=1 (p-state) cos parity = -1 l=2 (d-state) (3cos2-1) parity = +1 Spherical harmonics have (-1)l parity.
In its rest frame, the initial momentum of the parent nuclei is just its spin: Iinitial = sX and: Ifinal = sX'+ sa + ℓa 1p1/2 1p3/2 1s1/2 4He So |sX'– sX| < ℓa< sX'+ sX Sa = 0
Since the emitted a is described by a wavefunction: the parity of the emitted a particle is(-1)ℓ Which defines a selection rule: restricting us to conservation of angular momentum and parity. If P X' = P X then ℓ= even If P X' = -P X then ℓ= odd
|sX'– sX| < ℓa< sX'+ sX If the 2p, 2n not plucked from the outermost shells (though highest probability is that they are) then they will leave gaps (unfilled subshells) anywhere: Excited nuclei left behind! EXAMPLE: ℓa=sX' If SX = 0 (conservation of angular momentum) so PX' = -PX ℓa=3 0 3 nuclear transition would mean i.e.0+3- is possible, but 0+3+ is NOT possible 0 2 so PX' = PX do not change the parity of the nucleus 0 4 So 0+2- 0+4- would both be impossible