Lipids
E N D
Presentation Transcript
Lipids Lipids of Physiologic Significance
BIOMEDICAL IMPORTANCE • The lipids • The common property • insoluble in water • soluble in nonpolarsolvents • They are important dietary constituents • high energy value • fat-soluble vitamins • essential fatty acids • Fat is stored in adipose tissue
BIOMEDICAL IMPORTANCE • Thermal insulator • electrical insulators • cellular constituents • Membranes • Transport • Lipoproteins • Lipid biochemistry is necessary • obesity, diabetes mellitus, atherosclerosis, polyunsaturated fatty acids in nutrition and health
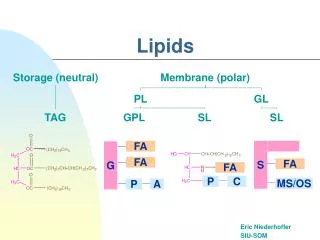
Classification • Simple lipids • Esters of fatty acids with various alcohols • Complex lipids • groups in addition to an alcohol and a fatty acid • Phospholipids • Glycerophospholipids • Sphingophospholipids • Glycolipids • Other complex lipids • Sulfolipids and aminolipids • Lipoproteins
Classification • Precursor and derived lipids • Neutral lipids • acylglycerols (glycerides),cholesterol, and cholesteryl esters
Free fatty • Free fatty acids • Saturated • -anoic • Unsaturated • -enoic • 1,2,3,… • α, β, γ ,… ω or n carbons • Δ
Unsaturated Fatty Acids • Monounsaturated • Polyunsaturated • Eicosanoids • eicosa- (20-carbon) polyenoic fatty acids • Prostanoids • Prostaglandins(PGs) • Prostacyclins (PGIs) • Thromboxanes (TXs) • Leukotrienes (LTs) • Lipoxins (LXs)
Prostaglandins • The number of double bonds • PG1, PG2, PG3 • Different substituent groups (on ring) • A, B,C,etc
The leukotrienes andlipoxins • Lipoxygenase pathway • three or four conjugated double bonds • Leukotrienes cause bronchoconstriction as well as being potent proinflammatoryagents and play a part in asthma
Unsaturated Fatty Acids • Geometric isomerism • Cis • on the same side • Trans • if on opposite sides • as a by-product of the saturation of fatty acids • ingestion of ruminant fat • Significance • Molecular packing in membranes
Unsaturated fatty acids of physiologic and nutritional significance
Structure of some unsaturated fatty acids
Physical and Physiologic Propertiesof Fatty Acids • Chain Length and Degree of Unsaturation • even-numbered-carbon fatty acids • The melting points
TRIACYLGLYCEROLS (TRIGLYCERIDES) • THE MAIN STORAGE FORMS OF FATTY ACIDS • Mono- and diacylglycerols
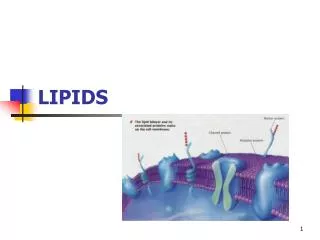
PHOSPHOLIPIDS • MAIN LIPID CONSTITUENTS OF MEMBRANES • derivatives of phosphatidic acid • Phosphatidylcholines (Lecithins) • the most abundant phospholipids of the cell membrane • represent a large proportion of the body’s store of choline
PHOSPHOLIPIDS • Surfactant • Dipalmitoyl lecithin • respiratory distress syndrome • Phosphatidylethanolamine (cephalin) • Phosphatidylserine • Phosphatidylinositol • Cardiolipin
Phospholipids • Plasmalogens
GLYCOLIPIDS • GLYCOSPHINGOLIPIDS • NERVE TISSUES • CELL MEMBRANE • Galactosylceramide • Sulfatide • Sulfogalactosylceramide • Glucosylceramide • Glycosphingolipid of extraneural tissues
Simple glycosphingolipids • Complex glycosphingolipids • Gangliosides • Sialic acid • The simplest ganglioside found in tissues is GM3 • Functions • Receptor, …
STEROIDS • Cholesterol • Atherosclerosis • As Precursor • Sterol • Has one or more hydroxylgroups and no carbonyl orcarboxyl groups
Stereoisomers • “Chair” or a “boat” form • The rings can be either cis or trans • β bonds • above the plane of the rings • α bonds • bonds attaching groups below
Cholesterol • Cholesteryl ester
Ergosterol • Precursor of Vitamin D
Polyprenoids • Synthesized from five-carbon isoprene units • Include • Ubiquinone • Dolichol • Vitamins • A, D, E, and K, and β-carotene (provitamin A)
LIPID PEROXIDATION • A SOURCE OF FREE RADICALS • Lipids exposed to oxygen • deterioration of foods (rancidity) • damage to tissues • Cancer, inflammatory diseases, atherosclerosis, and aging • Peroxide formation from fatty acids containing methylene-interrupted double bonds • Antioxidants • Vitamin E, Beta-carotene • Urate and vitamin C
AMPHIPATHIC LIPIDS • Form • Membranes, Micelles, Liposomes, & Emulsions • Contain polar groups • Fatty acids, phospholipids, sphingolipids, bile salts, and, to a lesser extent, cholesterol • Part of the molecule is hydrophobic, and part is hydrophilic
Micelles • When a critical concentration of these lipids is present in an aqueous medium • Facilitating absorption of lipids • Liposomes • Formed by sonicating an amphipathic lipid in an aqueous medium • as carriers of drugs
Emulsions • Much larger particles
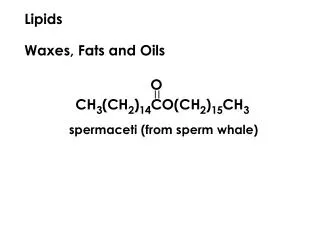
Waxes • Serve as • Energy Stores and Water Repellents • Esters of long-chain (C14 to C36) saturated and unsaturated fatty acids with long-chain (C16 to C30) alcohols • Their melting points (60 to 100 °C) are generally higher than those of triacylglycerols