What is the difference between accuracy and precision?
Warm Up (8/8/13). What is the difference between accuracy and precision?. Accuracy is being correct, Precision is being consistent. Unit 1 : Measurements, Graphing, 1-Dimensional Motion. Accuracy 101 – Accuracy vs. Precision.


What is the difference between accuracy and precision?
E N D
Presentation Transcript
Warm Up (8/8/13) What is the difference between accuracy and precision? Accuracy is being correct, Precision is being consistent.
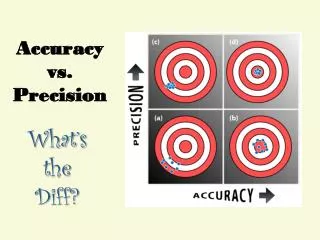
Accuracy 101 – Accuracy vs. Precision • The main goal when completing labs or any other task in chemistry is to be accurate! • Accuracy – the “correctness” or how close your data is to the right value.
Accuracy vs. Precision • Accuracy – the “correctness” of the data Precision – the consistency of the data. *Precision means being able to repeat results*
Accuracy vs. Precision • Determine if the trials below are accurate, precise, neither or both. Grades on 1st Semester Final Class Average: 80 Neither Accurate nor Precise Both Accurate and Precise Precise but not Accurate Accurate but not Precise
How do we accurately use extremely large and small numbers in calculations? • Example: What is 200000000 x .000000005? Answer: 1
Scientific Notation • Scientific Notation – illustrating a very large or small number using: 1) A number between 1 and 9.99 (single digit) 2) x 10 3) an exponent (positive or negative) Example: 3x108 and 6.022 x 10-23
Scientific Notation • How to convert a number in to scientific notation: • Find the decimal in the number. Ex. 300000000. 2) Move the decimal to make a single digit number. Ex. 300000000. 3) Determine the spaces and direction moved. Ex. 8 spaces to the left. 4) Write the single digit number with the x 10 Ex. 3.0 x 10 5) Write the exponent, positive if moved to the left, negative if moved to the right. Ex. 3.0 x 108
Scientific Notation • Examples: Convert the following number to scientific notation: .00000025 7 Spaces to the Right 2.5 x 10? 2.5 x 10-7 4000 3 Spaces to the Left 4.0 x 10 ? 4.0 x 10 3
Scientific Notation • Practice: Convert the following numbers into scientific notation. • 62300000 • .0000472 • 490100 • .000000080 • 72000 • .59 • 4.8 6.23 x 107 4.72 x 10-5 4.901 x 105 8.0 x 10-8 7.2 x 104 5.9 x 10-1 4.8 x 100
Scientific Notation • Practice: Convert the following numbers into standard form. • 5.4 x 102 • 9.21 x 10-6 • 2.7 x 1010 • 1.1 x 10-1 • 9.09 x 10-4 • 2.3 x 105 • 3.3 x 100 540 .00000921 27000000000 .11 .000909 230000 3.3
Scientific Notation • When typing Scientific notation into your calculator, you can enter it two ways: • Type the E or (EE) to replace the x 10 part. Example: 4 E 5 2) Type the x 10 and then use a carrot button (^) to enter in the exponent: Example: 4 x 10 ^ 5
Warm Up (8/9/13) Convert the following numbers into scientific notation: 35000000 .00000817 3.5 x107 8.17 x10-6
Dealing with large and small numbers II • The Metric System allows us to change the unit of measurement to a more manageable number. Example 10000000 mm = 10Km
Benefits to the Metric System • Based on the number 10 2) Universal 3) Can compare large and small numbers.
The Metric Scale K H D b d c m ing died hocolate enry by rinking ilk
The Metric Scale K H D b d c m ilo eca enti ecto ase eci illi
The Metric Scale K H D b d c m x 1000 X 10 x 100 g L s m ÷10 ÷ 1 0 0 ÷ 1 0 0 0
Base Units • Base units are the standard unit associated with different measurements: Time – Seconds (s) Length – Meters (m) Mass – grams (g) Volume – Liters (L) Force - Newtons (N) Power – Watt (w)
How to use the metric scale: The step – method. • Find the starting unit. • Move the steps to the ending unit. • Count spaces and direction moved • Move decimal same direction and spaces. Example: 12cm = Km K H D o d m .00012 C start finish
Examples of Metric Conversions 145000 1450. 1450 Dg = dg K H D o d c m .2750 Hm = m K H D o d c m start finish 27.50 .2750 start finish
Examples of Metric Conversions 2730 2.73 2.73 g = mg K H D o d c m .380 Km = m K H D o d c m finish start 380. .380 start finish
Practice: • Solve the following metric conversions. • 40cm = dm • 2.7KL = mL • 4.2 Ds = ds • .002 Hm = m • 78.5 cm = Km • 10.1 Hg = Dg • .45ms = Ks • 690 mg = g 4.0 2700000 420 .2 .000785 101 .00000045 .690
Significant Figures (Sig. Figs) • All known digits based on measurement tool.
Sig. Figs Rules 1) All non-zero numbers are considered significant figures. • Ex. 2.345 = 4 Sig. Figs 178,456.32 = 8 Sig. Figs 2) All zeros between non-zero numbers are considered significant figures • Ex. 20045 = 5 Sig. Figs 3007004 = 7 Sig. Figs
Sig. Figs Rule Cont. 3) All final zeros to the right of the decimal are considered significant figures. • Ex. 6.20 = 3 Sig. Figs .640000 = 6 Sig. Figs 4) Zeros that act as placeholders are not significant (convert to scientific notation) • Ex. 000235 = 3 Sig. Figs .000046 = 2 Sig. Figs
Significant Figures: Map Help Atlantic Ocean Pacific Ocean Decimal Absent Decimal Present *Continue until you hit any number other than zero. Anything crossed out is not significant*
Significant Figures: Map Help *There are only 4 significant figures in this number.* Atlantic Ocean 4301000 Decimal Absent *Continue until you hit any number other than zero. Anything crossed out is not significant*
Significant Figures: Map Help Pacific Ocean *There are only 3 significant figures in this number.* .000250 Decimal Present *Continue until you hit any number other than zero. Anything crossed out is not significant*
Significant Figures: Map Help *There are only 5 significant figures in this number.* Atlantic Ocean 1240100 Decimal Absent *Continue until you hit any number other than zero. Anything crossed out is not significant*
Significant Figures: Map Help Pacific Ocean *There are only 6 significant figures in this number.* 0.0450000 Decimal Present *Continue until you hit any number other than zero. Anything crossed out is not significant*
Practice Sig. Fig Problems • 48.30 = S.F. • 00023004. = S.F. • 67000 = S.F. • .00050 = S.F. • 4.04000 = S.F. • 34.002 = S.F. • .000700 = S.F. 4 5 2 2 6 5 3
Warm Up (8/12/13) Which of the following numbers would be considered the most precise? 215 5000000 .0004200 3S.F. 4 S.F. 1 S.F. Most Precise
Dimensional Analysis“The Bridge Method” Dimensional Analysis – method used to convert units based on the ratio of conversions. *Works for both base and derived units!* Base Unit – One unit (Ex. Km, g, second) Derived Unit – Two units working together to form one measurement (Ex. Density g/mL)
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 1 – Write down what you start with the units on top of each other (like a fraction). *Hint: Any whole number is understood to be divided by 1. 200g mL 1
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 2 – Draw a “block” of your bridge next to your starting number. *Hint: Do not draw more until you know that you need them. (Some can be done in one step). 200g mL 1
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 3 – Pick a unit (doesn’t matter which one) then write the unit in the opposite spot from where it is (this will cancel the unit!). Then place the unit you want to go to in the connecting box. 200g kg mL 1 g
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 4 – Fill in the appropriate ratio for the units. *Hint: The bigger unit will always have a value of 1 and the smaller unit will be bigger than 1 to account for all the smaller units that make up the larger units. 200g 1 kg mL 1 1000 g
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 5 – Repeat steps 2-4 until all units have been adjusted to the correct units. *Hint: Sometimes it easier to use an intermediate (more than one) conversions for one unit. 200g 1 kg 10 mL mL 1 1000 g cL 1
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 6 – Cross out any units that are on the top and bottom of the bridge. *Hint: The only units left should be the correct units based on the question. 200g 1 kg 10 mL mL 1 1000 g cL 1
How to use the “Bridge Method” A cube with a density of 200 g/mLhas a density of how many kg/cL? Step 7 – Multiply the numbers on the top and then multiply the numbers on the bottom. Then divide the top by the bottom. *Hint: Ones can be left out as they do not effect the final calculation. 200g 1 kg 10 mL (200 x 1 x 10) 2000 = mL 1 1000 g (1 x 1000 x 1) 1000 cL 1 2 kg/cl
How to use the “Bridge Method” Example: How many seconds are in a 1 year? 1 year 24 hours 60 min. 365 days 60 sec. 1 days 1 hours min. 1 year 1 1 x 365 x 24 x 60 x 60 31536000 = 1 x 1 x 1 x 1 1 31,536,000 seconds in a year
How to use the “Bridge Method” Example: How many cm are in a 4 km? 1000 4 km 100 cm m 1 m 1 km 4 x 1000 x 100 400000 = 1 x 1 1 400000 cm in 4 km
How to use the “Bridge Method” Example: How many kg/L are in 2.5 g/mL? 2.5 g 1000 1000 mL 1 kg mL 1 L g 2.5 x 1 x 1000 2500 = 1 x 1000 x 1 1000 2.5 kg/L in 2.5 g/mL
How to use the “Bridge Method” Example: How many Dm/s are in 9m/ms? 9 m 1000 ms 1 Dm 1 ms 1 s 10 m 9 x 1 x 1000 9000 = 1 x 10 x 1 10 900 Dm/s in 9 m/ms
How to use the “Bridge Method” Example: How many cg/L are in 20g/mL? 1000 20 g 100 cg mL 1 mL 1 g L 1 20 x 1000 x 100 2000000 = 1 x 1 x 1 1 2000000 cg/L in 20 g/mL
How to use the “Bridge Method” Example: How many m/s are in 55Km/hr? 55 Km 60 Hr 1 1 min. 1000 m min 60 sec 1 Hr 1 Km 55 x 1000 x 1 x 1 55000 = 1 x 1 x 60 x 60 3600 15 m/s in 55 Km/hr
Warm Up (8/12/13) Which of the following numbers would be considered the most precise? 215 5000000 .0004200 3S.F. 4 S.F. 1 S.F. Most Precise
Warm Up (8/13/13) How many m/s are in 14Km/hr? 14 Km 60 Hr 1 1 min. 1000 m min 60 sec 1 Hr 1 Km 14 x 1000 x 1 x 1 14000 = 1 x 1 x 60 x 60 3600 3.9 m/s in 14 Km/hr