RADIOACTIVE DATING
RADIOACTIVE DATING. CH 10-3. GEOLOGIC DATING: ABSOLUTE AGE DETERMINATION. Radioactivity was first discovered by Henri Becquerel in 1896 and Polish-French chemist Marie Curie discovered that radioactivity produced new elements ( radioactive decay ).

RADIOACTIVE DATING
E N D
Presentation Transcript
RADIOACTIVE DATING CH 10-3
GEOLOGIC DATING: ABSOLUTE AGE DETERMINATION • Radioactivity was first discovered by Henri Becquerel in 1896 and Polish-French chemist Marie Curie discovered that radioactivity produced new elements (radioactive decay). • Ernest Rutherford first formulated the law of radioactive decay and was the first person to determine the age of a rock using radioactive decay methods. Marie Curie Ernest Rutherford
GEOLOGIC DATING: ABSOLUTE AGE DETERMINATION • The number of protons (the atomic number) is fixedfor any element and is unique for each element but the number of neutrons in atoms of different elements can vary. Atoms of an element having different numbers of neutrons are referred to as the isotopes (of that element). M&W4 Fig. 3.3; M&W5 Fig. 3.4
GEOLOGIC DATING: ABSOLUTE AGE DETERMINATION Radioactive decay occurs when an isotope of one element is transformed into a different element by changes in the nucleus. There are three different decay mechanisms: “Parent” “Daughter” M&W4 Fig. 17.18; M&W5 Fig. 17.18
Radioactive Dating When the atoms of one element break down to form atoms of another element.
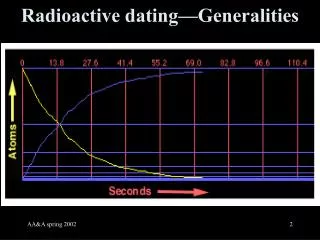
How can we tell age based on the number of parent isotopes? Radioactive isotopes “decay” at a particular rate. We express this rate as the “HALF-LIFE”, which is the time it takes for HALF of the parent isotopes to decay.
Half Life The amount of time it takes for half of the radioactive atoms to decay.
GEOLOGIC DATING: ABSOLUTE AGE DETERMINATION • For radioactivity dating we use igneous rocks and minerals. The clock starts when radioactive atoms that are present in the magma get incorporated in the crystalline structure of certain minerals in the rocks. • The crystals containing the parent atoms formand so we then have a “container” with parents that can begin decaying to form daughters. • We can then use measure the parent-daughter ratio. This is our “atomic clock” that records the time since the rock crystallized. M&W4 Fig. 17.21; M&W5 Fig. 17.21
GEOLOGIC DATING: ABSOLUTE AGE DETERMINATION • To the oldest materials ever dated by the radioactive method are found in the Jack Hills of western Australia and are tiny zircon grains contained in sandstones and conglomerates. The zircons are 4.4 billion years old. The very remote “outback” of western Australia--the Jack Hills Scanning electron microscope image of a Jack Hills zircon. Scale bar is 0.1 mm
Examples of Radioactive Changes in Rock • Carbon 14 - 5,730 years changes to Nitrogen • Potassium 40 – 1,3 billion years changes to Argon • Uranium 238 – 5.5 billion years changes to Lead
C14 is an isotope of carbon that forms from Nitrogen in the atmosphere. Living things consume this radioactive carbon. Once dead, no new carbon is absorbed, and C14 turns back into Nitrogen. The Half-Life of C14 is 5,730 years. This method works best for fossils younger than 50,000 years. Why? (end) M&W4 Fig. 17.24; M&W5 Fig. 17.24