Radioactive dating
Radioactive dating. Radioactive Dating.

Radioactive dating
E N D
Presentation Transcript
Atoms are the basic building blocks of matter, in the same way that bricks are the basic building blocks of a house. Atoms are incredibly small, too small even to be seen with a microscope. There are 9 sextillion atoms (9,000,000,000,000,000,000,000) in a penny.
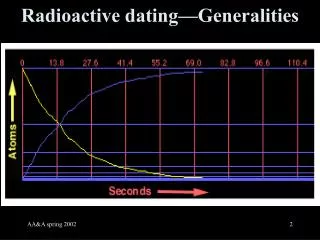
Most atoms are STABLE- they do not change • Some atoms are unstable – they are said to be radioactive. • During radioactive decay, the atoms of one element break down to form atoms of another element
ISOTOPES • Often, isotopes are unstable and thus are radioactive. • Isotopes are atoms that have a different number of neutrons than normal. • C-12 is stable • C-14 has two more neutrons than normal and is unstable/radioactive. It gives off radiation and particles until it becomes stable.
During radioactive decay, the atoms of one element break down to form atoms of another element.
Half Life • Half life is the amount of time is takes half of a radioactive element to decay (break down) into a stable (nonradioactive) element. • Ex: p. 333 • The half life of Carbon 14 is 5730 years. • If you began with 100 grams of Carbon 14, it would take 5730 years for half of that sample to decay. After 5730 years (one half life), you would have 50 grams of Carbon 14 left and 50 grams of a stable element.
The original material is called the parent material. What is decays into is called the daughter material.
Example • Dating a Fossil “Radioactive Isotopes and Fossil Age” • Start with 100 grams of a certain radioisotope that has a half life of 50,000 years. This means that half of the radioactive material becomes stable (non radioactive) after _________ years.
The Iceman • Carbon dating is used to find the age of once living organisms. • It is based on the amount of radioactive carbon (C-14) found in the organism • The half life of C-14 is 5730 years. • They found ¼ as much as C-14 in the iceman as is normally found in living organisms. • How old did they determine him to be?
PRACTICE • RADIOACTIVE DATING ENRICHMENT ACTIVITY • KNOW THESE TERMS FOR TOMORROW: • MATTER • ATOMS • ISOTOPE • ELEMENT • STABLE • DECAY • HALF LIFE • CONSTANT • SECTION 3 ASSESSMENT P334 CORNELL NOTES ALL (INCLUDING MATH PRACTICE)