Fundamentals of Atomic Structure and Chemical Bonding
Understand the basics of atomic structure, isotopes, electron configurations, Lewis structures, electronegativity, resonance structures, and common bonding patterns in this introductory chapter review.

Fundamentals of Atomic Structure and Chemical Bonding
E N D
Presentation Transcript
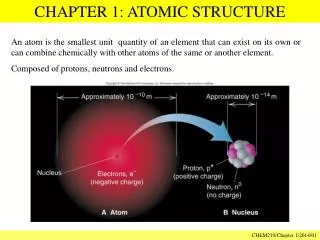
ATOMIC STRUCTURE • number of protons determines the element • combinations are possible, with a constant number of protons but varying number of neutrons - ISOTOPES Chapter 1: Introduction and Review
ORBITALS AND SYMMETRY Chapter 1: Introduction and Review
THE BUILDING OF THE ELECTRONIC CONFIGURATION OF ATOMS • A maximum of two (2) electrons can be placed in any single orbital (The Pauli’s exclusion principle) • Electronic states are occupied in order of increasing energy. • Degenerate states (of equal energy!) are first all single occupied (The Hund’s rule).
Lewis’ Octet Hypothesis • Atoms will enter into chemical reactions and form bonds with the principle goal to achieve octet configurations of their valence shells
ELECTRONEGATIVITY – PAULING’S SCALE Chapter 1: Introduction and Review
RULES FOR LEWIS STRUCTURES • Count the total number of electrons available (A) • Calculate the total number of necessary electrons (B), in • order for each atom to achieve an octet (Hydrogen needs • only two electrons!!) • Subtract and divide: (B – A)/2 = number of bonds • Construct a meaningful structure with the above calculated • number of bonds • If electrons remain, first assign lone pairs to the terminal • atoms, then to the central atoms, wherever necessary • Do not forget: Hydrogens are ALWAYS terminal! Chapter 1: Introduction and Review
RESONANCE STRUCTURES • A resonance structure is acceptable if: • it is a meaningful Lewis structure (Please refer to rules for writing Lewis structures!) • electrons are moved but the skeleton of the molecule is retained • A resonance structure has a greater importance if: • it contains a greater number of bonds and atom octets • it has minimum charge separation • the negative charge resides on a more electronegative atom Chapter 1: Introduction and Review
COMMON BONDING PATTERNS Chapter 1: Introduction and Review