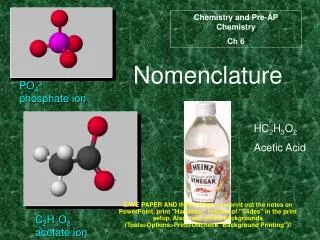
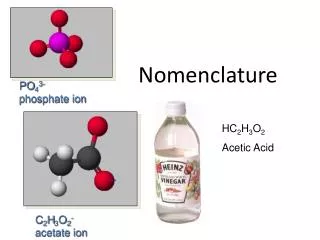
Understanding Acid Nomenclature: Binary and Oxyacids Explained
This guide provides a comprehensive overview of acid nomenclature, focusing on both binary acids and oxyacids. For binary acids with the formula HX, the naming convention involves adding "Hydro" to the root of the anion name and ending with "ic acid" (e.g., HCl becomes hydrochloric acid). Oxyacids, derived from polyatomic ions, follow different rules based on the suffix of the anion: “-ate” becomes “-ic acid” while “-ite” becomes “-ous acid”. Examples, writing formulas, and key terms are also covered.

Understanding Acid Nomenclature: Binary and Oxyacids Explained
E N D
Presentation Transcript
Acid Nomenclature • Binary Acids • HX hydrogen _____ide • becomes • Hydro______ ic acid
HCl(g) hydrogen chloride • HCl (aq) hydrochloric acid • H2S(g) hydrogen sulfide • H2S(aq) hydrosulfuric acid • HCN(g) hydrogen cyanide • HCN(aq) hydrocyanic acid
Oxyacid Nomenclature • Hydrogen ___ate ___ic acid • Hydrogen ___ite ___ous acid • Hydrogen per __ate per__ic acid • Hydrogen hypo__ite hypo__ous acid
H2SO4 H2SO3 H2CO3 HClO H2TeO3 HBrO4 Examples
H2SO4 H2SO3 H2CO3 HClO H2TeO3 HBrO4 sulfuric acid sulfurous acid carbonic acid hypochlorous acid tellurous acid perbromic acid
hydro Writing the Formulas for Acids FIRST You must determine the formula for the anion, “(X)”. • If the acid’s name starts with “________” (and ends in “–ic”), the name of the anion used ends in “_______”. Therefore, it is a _________ acid. (There’s only hydrogen and one other nonmetal in the formula!) • The subscript on the hydrogen equals the anion’s charge! Examples: hydrobromic acid = ________ hydroiodic acid = _______ hydrosulfuric acid = ________ -ide binary HBr HI H2S
-ic -ate oxy- • If the acid’s name ends with “______” without the “hydro-” prefix, the name of the anion used ends in “_______”. Therefore, it is an ____ acid. • The subscript on the hydrogen equals the anion’s charge! Examples: perchloric acid = __________ oxalic acid = ___________ • If the acid’s name ends with “______”, the name of the anion used ends in “_______”. (It’s is also an oxy-acid.) • The subscript on the hydrogen equals the anion’s charge! Examples: hypochlorous acid = __________ nitrous acid = ____________ sulfurous acid = ____________ HClO4 H2C2O4 -ous -ite HClO HNO2 H2SO3
Examples • Chromic acid • Hydroiodic acid • Chlorous acid • Perchloric acid • Carbonic acid • Phosphorous acid • Hydroselenic acid
Examples • Chromic acid • Hydroiodic acid • Chlorous acid • Perchloric acid • Carbonic acid • Phosphorous acid • Hydroselenic acid • H2CrO4 • HI • HClO2 • HClO4 • H2CO3 • H3PO4 • H2Se