The Polyatomic Ions
DESCRIPTION
The Polyatomic Ions. You Need to know!!!!!!. NH 4 +. Ammonium. H 3 O +. Hydronium. OH –. Hydroxide. CN –. Cyanide. SCN –. Thiocyanate. NO 3 –. Nitrate. NO 2 –. Nitrite. CO 3 2 –. Carbonate. HCO 3 –. Bicarbonate (aka: Hydrogen Carbonate). C 2 O 4 2–. Oxalate. S 2 O 3 2–.
1 / 24
Download Presentation


Télécharger la présentation 

The Polyatomic Ions
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
The Polyatomic Ions You Need to know!!!!!!
NH4+ Ammonium
H3O+ Hydronium
OH– Hydroxide
CN– Cyanide
SCN– Thiocyanate
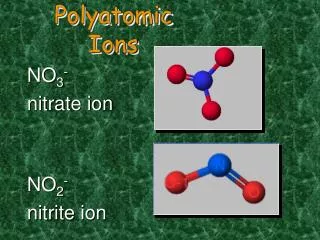
NO3– Nitrate
NO2– Nitrite
CO32– Carbonate
HCO3– Bicarbonate (aka: Hydrogen Carbonate)
C2O42– Oxalate
S2O32– Thiosulfate
SO42– Sulfate
SO32– Sulfite
PO43– Phosphate
PO33– Phosphite
ClO– Hypochlorite
ClO2– Chlorite
ClO3– Chlorate
ClO4– Perchlorate
C2H3O2– orCH3COO– Acetate
MnO4– Permanganate
CrO42– Chromate
Cr2O72– Dichromate
More Related

Audio
Live Player