Chapter 4. Biological Oxidation
Chapter 4. Biological Oxidation. Intredction ATP oxidative phosphorylation Oxidation not producing ATP. introduction biological oxidation

Chapter 4. Biological Oxidation
E N D
Presentation Transcript
Chapter 4. Biological Oxidation Intredction ATP oxidative phosphorylation Oxidation not producing ATP
introduction biological oxidation oxidation run in living body, detailedly, the process which nutrient substance, such as saccharides, lipids, and proteins are oxidized into water and carbon dioxide, and simultaneouslyproduce energy. Nutrient + O2 H2O + CO2 + energy ATP + heat
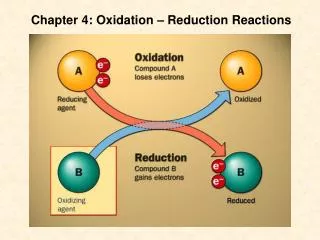
oxidizing reaction loss of electrons dehydrogenation addation of oxygen reduction reaction Gain eletrons Addation of hydrogen Deoxygenation +H +e [O] [R] -e -H +O Forms ofoxidoreductionin biological oxidation
glucose fatty acid + Glycerol amino acids General course of biological oxidation glucogen triglyceride protein AcetylCoA TAC ADP+Pi ATP CO2 H2O 2H respiratory chain
Energy of reaction G < 0 spontaneous G = 0equilibrium G > 0non-spontaneous G1 G2 G = G2-G1 G0’= - 2.303 R T log Keg = - n F E0’
adenosine triphosphoric acid 腺苷三磷酸 Section 1. ATP
ATP is the main form of energy utilization and store in body and the center of energy conversion.
GDP GTP ATP + UDP ADP + UTP CDP CTP Transformbetween high-energy compounds nucleoside diphosphate kinase adenylate kinase ADP + ADP ATP + AMP
Section 2. oxidative phosphorylation • Ways producing ATP: • substrate level phosphorylation • oxidative phosphorylation substrate level phosphorylation --formation of ATP by the way of straight transfer high-energy substrate energy to ADP.
1,3-二磷酸甘油酸 + ADP 3-磷酸甘油酸 + ATP 磷酸烯醇式丙酮酸 + ADP 丙酮酸 + ATP 琥珀酰CoA + H3PO4 + GDP 琥珀酸 + CoA-SH + GTP + ADP ATP PK
I respiratory chain (呼吸链) aoxidoreduction system which consists of a series of enzyme, coenzyme aligning in mitochondrial inner membrane, function as linksystem transferer of hydrogen and electron.
Mitochondria Respiratory chain
Cytc 内外膜间隙侧 Q I Ⅲ Ⅳ 线粒体内膜 Ⅱ 基质侧 electron transfer in respiratory chain e- e- e- e- e- H2O NADH+H+ 延胡素酸 1/2O2+2H+ NAD+ 琥珀酸
succinic acid oxidation respiratory chain NADH oxidation respiratory chain
Component of respiratory chain Co-Q and Cyt C
NADH FMN,Fe-S CoQ (1) complexⅠ— NADH-Q reducase • iron-sulphur protein, Flavoprotein with FMN, 42 peptide chains, 850 kD. • Bind and oxidize NADH, transfer electrons to Q, release 4H+ to interspace of inner and outer membrane.
Structure of NAD+ and NADP+ R=H:NAD+; R=H2PO3:NADP+ NAD+:nicotinamide adenine dinucleotide,CoI NADP+:nicotinamide adenine dinucleotide phosphate,CoII
Fe2+ Fe3+ + e Fe4S4 ironsulfur protein Fe-S Fe2S2, Fe4S4
Ubiquinone,Q (Coenzyme Q,CoQ) 人体中: CoQ10 • quinones contain a polyisoprene side chain. • liposolubility,make it move in mitochondrial inner membrane easily. • the only one electron carrier without protein in respiratory chain.
2H+ FMN Fe-S N-2 Q QH2 复合体Ⅰ传递电子的过程 2e- 2H+ NAD+ NADH+H+ NADH+H+ FMN Fe2+ Q FMNH2 Fe3+ QH2 NAD+ Q NADH FMN Fe-S
(2) complexⅡ- Succinate-CoQ reductase • i.e. succinate dehydrogenase, consists at list 4 peptides. Contain one FAD, two ironsulfur protein and one Cyt b560. • Transfer electron from succinic acid to Q, do not release H+ to the interspace. succinic acid→FAD→Fe-S→Q。
cytochrome,Cyt 细胞色素 A、structure: colourant protein containing iron porphyrin. B、typing: Cyta: Cytaa3 Cytb: Cytb562、Cytb566、 Cytb560 Cytc: Cytc 、 c1 C、difference: ① different side chain of iron porphyrin. • Different linkage form of iron porphyrin with the protein. CytFe3+ + e CytFe2+
多聚异戊二烯长链 甲酰基
(3) Complex Ⅲ Q-cytc reducase • i.e. cyt c reducase, consists of 11 peptide chains different, existing as a dimer. every monomer contains two cyt b (b562, b566), one cyt c1 and a iron sulphur protein. • Catalyze electron transfer from Q to cyt c. every two electrons’s transfering lead four proton pumped to the intermembrance space. Cytc QH2 b566b562Fe-Sc1
Cyt c Complex Ⅲ
Electron transfering process in complexⅢ first time oxidation of QH2 secondary time oxidation of QH2 Cytc Cytc 2H+ 2H+ Cytc1 Cytc1 Fe-S Fe-S e- e- bL bL e- e- QH2 Q QH2 Q bH bH Q Q QH2 Q 2H+
(4) complex Ⅳ Cyt c oxidase • Dimer. Every monomer consists of 13 peptide chains different, as 3 subunits: I include 2 heme(a,a3),a cuproprotein (CuB);Ⅱinclude a dikaryon center formed by two copper ion(CuA);Ⅲ not clear • Cu2+ + e Cu+ Cyt c CuA a a3 CuB O2
NADH氧化呼吸链 NADHFMN(Fe-S)Qbc1caa3O2 琥珀酸氧化呼吸链 succinic acidFAD(Fe-S)Qbc1caa3O2
呼吸链 NADH + H+ + 1/2O2 NAD+ + H2O 呼吸链 FADH2 + 1/2O2 FAD + H2O 呼吸链 2H + 1/2O2 H2O In FADH2 or NADH overall reaction or further
呼吸链中电子传递体的排列顺序的确定 • 利用脱氧胆酸处理线粒体内膜、分离出呼吸链的4种复合物,辅酶Q和细胞色素C及ATP合酶。 • 根据标准氧还电位E0’的高低排列 • 根据电子传递体氧化还原态时的吸收光谱变化进行检测 • 利用阻断剂研究分析 • 四种复合物的电子传递再造实验
II、oxidative phosphorylation oxidative phosphorylation refer to the ATP producing form which the reaction ADP change into ATP couple withrespiratory chain oxidation. It is main form of ATP producing in body.
energy ATP+H2O 30.5kJ/mol ADP + Pi energy level diagram Why does the reaction: ADP + Pi ATP + H2O request a couple? The product state(ATP+H2O) is higher energy level than reactant state(ADP + Pi). So, ADP change into ATP isn’t spontaneous process. Energy obtaining is requested for the process.
Which segment in respiratory chain can produce enugh energy for ADP phosphorylation?
What’s P/O ratio?What’s its meaning? --- number of moles of ATP produced as consuming a mole of oxygen atom in a reaction, i.e. the number of moles of phosphor cosumed when consume a mole of oxygen atom in the reaction.
1961~1978 • What’s the way of ADPphosphorylation coupled with respiratory chain oxidation? 氧化磷酸化偶联机制有:化学物质偶联学说、构象偶联学说及化学渗透学说。目前公认度较大的是化学渗透学说。 Chemiosmotic hypothesis • the energy of respiratory chain oxidation change into proton gradient across the inner membrane. • the proton gradient drive ATP-synase produce ATP.
In electron transfer process, respiratory chain put proton to intermembrance of inner and outer membrance, result in proton concentration different of the two side of the inner membrance.
conceptual diagram of Chemiosmotic hypothesis H+ H+ H+ H+ H+ H+ + + + + + + + + + + + + + + + + + + + - - - - - - - - - - - - - - - e- 延胡素酸 H2O NADH+H+ 琥珀酸 1/2O2+2H+ NAD+ ATP ADP+Pi
ATP synase • Consists of hydrophobic F0(a1b2c912)and hydrophilic F1(33). • When proton go straight through a,push c loop turning , and as a result, spur the F1 turning.
松开 (O) (O) H+ 结好 (L) H+ 脱水 (T) H+ work principle of ATP synase Three conformations of subunit:松L,紧T,放O Procedure of ATP production:
Ⅲ. Factors affecting oxidation phosphorylation • Inhibitors • Regulation by ADP • Thyroid hormone • Mitchondrial DNA mutation
(1)Inhibitors • Inhibitors of respiratory chain blockelectron transfer of respiratory chain. • Uncoupler destroy the coupling of oxidation with phosphorylation, like uncoupling protein, 2,4-dinitrophenol. • Inhibitors of oxidative phosphorylation restrain the proton return to matrix side in ATP synase, like oligomycin