Aromatic Nitration - Mechanism
160 likes | 920 Vues
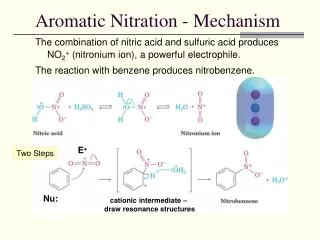
Aromatic Nitration - Mechanism. The combination of nitric acid and sulfuric acid produces NO 2 + (nitronium ion), a powerful electrophile. The reaction with benzene produces nitrobenzene. E +. Two Steps. Nu:. cationic intermediate – draw resonance structures. Aromatic Sulfonation.

Aromatic Nitration - Mechanism
E N D
Presentation Transcript
Aromatic Nitration - Mechanism The combination of nitric acid and sulfuric acid produces NO2+ (nitronium ion), a powerful electrophile. The reaction with benzene produces nitrobenzene. E+ Two Steps Nu: cationic intermediate – draw resonance structures
Aromatic Sulfonation Add a sulfonic acid group (-SO3H) to the ring using sulfuric acid and SO3. With –CF3, the new group is added meta. What is the electrophile?
Aromatic Sulfonation • Reaction with a mixture of sulfuric acid and SO3 gas (“fuming sulfuric acid”). • Electrophile is sulfur trioxide (SO3) or its conjugate acid. • Reaction occurs via cationic intermediate and is reversible.
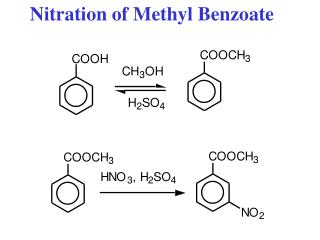
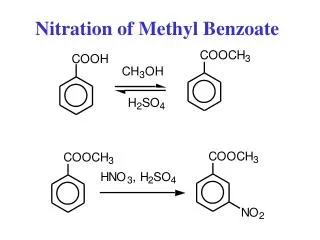
Review: EAS Reaction Conditions Halogenation (Cl2 or Br2): Benzene Nitration: Aromatic Sulfonation:
Effect of Substituents But what if the benzene ring has a substituent? Q1 Where does the new group go? Q2 Is the reaction faster or slower than benzene? x
Substituent Effects in Aromatic Rings Substituents can make a ring more reactive or less reactive: “activators” vs. “deactivators” Substituents also control orientation of the reaction:“ortho/para directors” vs. “meta directors” least reactive most reactive
Substituent Effects Q. How do different substituents influence the rate of electrophilic aromatic substitution reactions? activate the ring = faster reaction deactivate ring = slower reaction Q. How do different substituents effect orientation of the substitution reaction? (ortho/para vs. meta substitution) Answer: Inductive EffectsResonance Effects
Inductive vs. Resonance Effects Inductive EffectResonance Effect Based on: electronegativity conjugation Electrons Move: via sigma bonds pi bonds/lone pairs Distance: short-range may be long range Overall Effect: electron cloud pi bonds and is shifted lone pairs move by electronegativity by resonance Example: