Aromatic
Aromatic. Compounds. Good Aromatics. cinnamon. O ║. CH ═ CH – C – H . cinnamaldehyde. coffee. O ║. CH3. 3HC. N. N. O ║. N. N. CH3 . caffeine. vanilla. OH. OCH3. H – C ═ O. vanillin. Filipino BBQ goodness: puto (rice muffins). a niseed/anise. OCH3. CH ═ CH – CH3.


Aromatic
E N D
Presentation Transcript
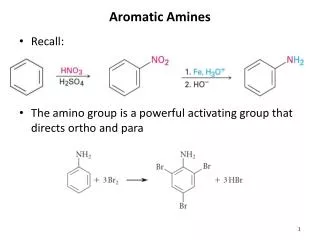
Aromatic Compounds
Good Aromatics
cinnamon O ║ CH ═ CH – C – H cinnamaldehyde
coffee O ║ CH3 3HC N N O ║ N N CH3 caffeine
vanilla OH OCH3 H – C ═ O vanillin
Filipino BBQ goodness: puto (rice muffins) aniseed/anise OCH3 CH ═ CH – CH3 ANETHOlE
cloves OH OCH3 CH2 – CH ═ CH2 eugenol
wintergreen O ║ C – O – CH3 OH Methyl salicylate
chamomile O ║ O COUMARIN
Bad Aromatics
benzene benzene
naphthalene nApHthalENE
burnt meat PHEnANTHRENE
smoke belchers BEnZOPYRENE
smoke belchers PYRENE CORONENE
cigarettes BEnZanthRacENE diBEnZanthRacENE
cigarettes NO N N nITROSONORNICOTINE
Lets try to find out the missing piece of the puzzle that leads to that occurrence…
“ Let us learn to dream, gentlemen, and then perhaps we shall learn the truth.” -1890 Kekulé -
Some of the early researchers in organic chemistry became intriguedby fragrant oils that could be extracted from certain plants. The compounds responsible for thearomas, had similar chemical properties. As a result, they weregroupedtogether and calledAROMATICS COMPOUNDS.
As more and more aromatic compounds wereisolated and studied, chemist gradually realized that aromatic contained six carbon atoms and had low hydrogen – to – carbon ratios (relative to other hydrocarbon). They also realized that the term aromatic was not always accurate. Because there are many fragrant compounds do not have aromatic properties.
1825 – Michael Faradaydiscovered BENZENE in whaleoil.When BENZENE was found to have the formula C6H6, scientist was puzzled.They tried unsuccessfully to draw a straight – line structure for BENZENE.
BENZENE’s possible structures CH3— C ≡ C ─ C ≡ C — CH3 2,4 – hexadiyne CH2═ CH ─ C ≡ C ─ CH ═ CH2 1,5 – hexadien – 3 – yne
Kekule was to say later that he must have dozed off at this point. In his dreamthe black balls of carbon turned into black impswith forked tails that began racing around the room and would soon be upsetting the apparatus of the laboratory. He was ready to run the rascals out. Then, almost suddenly, the confusion died away as each imp grabbed the tail of the one ahead of him, the six forming a whirling circle. One hand of each imp held a tail, the other a white handkerchief--and they waved to him as the group whirled by. He said that he came awake with a start, realizing that the imps were acting out the formula for benzene. As his hand grabbed the sketching pencil, the imps were back to black balls again and the handkerchiefs had changed to hydrogen atoms. How simple the arrangement turned out to be. “The carbon atoms of benzene form a ring."
1858 – Friedrich August von StradonitzKekulé.Suggested that the six carbon atoms formed a cyclic structure having three sets of alternating double bonds.1865 – Kekulé proposed that BENZENE had two structural formulas and that is alternated between them.
1931 – Linus Pauling To help explain molecules like benzene, he proposed resonance theory. (Pauling also gave us hybrid orbitals, electronegativity, and valence bond theory).That gave birth to the modern structure of BENZENE.
Rule # 1 • When a single hydrogen of the benzene ring is replaced, the compound can be named as a derivative of benzene.
Examples: Cl chlorobenzene CH2CH3 Br NO2 ethylbenzene nitrobenzene bromobenzene
Rule # 2 • A number of benzene derivatives are known by common names that are also IUPAC – accepted and are used preferentially over other possibilities.
Examples: CH3 NH2 O ║ OH C — OH Aniline Toluene Benzoic Acid Phenol
O ║ C – H EXAMPLES O ║ C – CH3 benzaldehyde acetophenone O ║ C benzophenone
Rule # 3 • Compounds formed by replacing a hydrogen of benzene with more complex hydrocarbon group can be named by designating the benzene ring as substituent. • We called them phenyl group. • benzyl
Examples: 3HC CH3CHCH ═ CH2 Br ا ا ا CH3— C — C — C — CH3 ا ا ا Br 3HC 3 – phenyl – 1 – butene 2,2 – dibromo – 4,4 – dimethyl – 3,3 – diphenylpentane
OH CH2 2 – benzyl – 4 – chlorophenol Cl
Rule # 4 • When two groups are attached to a benzene ring, three isomeric structures are possible. They can be designated by the prefixes ortho (o), meta (m), and para (p):
Examples: CH3 CO2H Cl Br NO2 O - bromochlorobenzene Cl m – nitrobenzoic acid p - chlorotoluene
Examples: CH3 CH3 CH3 CH3 CH3 1,2 – dimethylbenzene 1,4 - dimethylbenzene CH3 p - xylene O – xylene 1,3 - dimethylbenzene m – xylene
Rule # 5 • When two or more groups are attached to a benzene ring, their positions can be indicated by numbering the carbon atoms of the ring so as to obtain the lowest possible numbers for the attachment positions. • Group that comes first in alphabeticalorder is given the lower number.
Examples: CH3 Br Cl OH O2N NO2 Br NO2 I 3 – bromo – 5 - nitrotoluene 1 – bromo – 2 – chloro – 4 – iodobenzene NO2 2,4,6 – trinitrophenol