Aromatic compounds
Aromatic compounds. Benzene. Objectives. What are they?. Knowing what you know. Try to draw the atomic structure of Benzene C 6 H 6. Think about the number of bonds each carbon atom has!. Actual representations. Trienes.

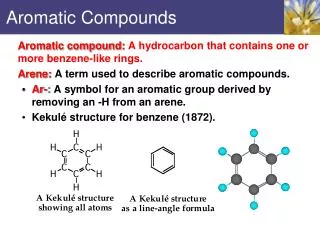
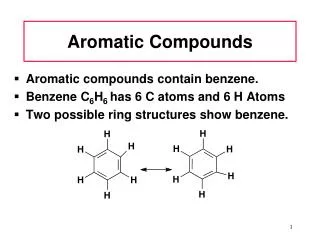
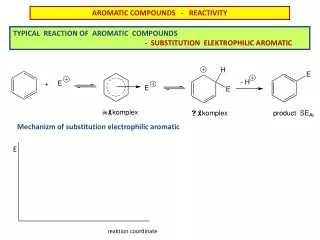
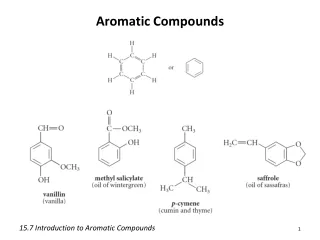
Aromatic compounds
E N D
Presentation Transcript
Aromatic compounds Benzene
Knowing what you know • Try to draw the atomic structure of Benzene C6H6. Think about the number of bonds each carbon atom has!
Trienes • Originally a scientist called Kekule suggested this structure for the benzene ring This is referred to as a triene Why?
But • Benzene and chemicals made from benzene rings joined together (conjugated) such as Naphthalene are much more stable than you’d predict. • At the time Kekule could not have explained this as electrons were not known
Modern representation After Robinson 1947 Referred to as cyclic conjugation In effect 6 πelectrons fly round the middle making it a very stable structure.
Benzene rings can be joined • By single bonds such as biphenyl:
Physical properties Like other hydrocarbons they are:
Marks • 20 marks per chemical (1 aromatic and 1 polyaromatic) • 10 marksfortheproduction of fiveclearunambiguousquestionsbasedonyour poster • Chemicaldividedintofivethemes, eachworth 4 marks. • 4= outstanding • 3 = good • 2 = satisfactory • 1 = poor • Total out of 50. • Good posters to be displayed in school.