AROMATIC COMPOUNDS
AROMATIC COMPOUNDS. Meeting “Aroma”. - Definition of aromatic molecule: a molecule containing one or more benzene rings. Identifying aromatic compounds. Aromatic compounds in our daily life. Aromatic ring (benzene ring).


AROMATIC COMPOUNDS
E N D
Presentation Transcript
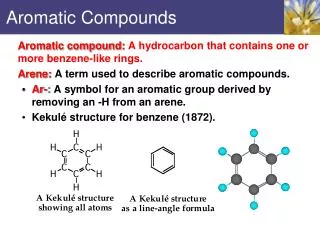
- Definition of aromatic molecule: a molecule containing one or more benzene rings.
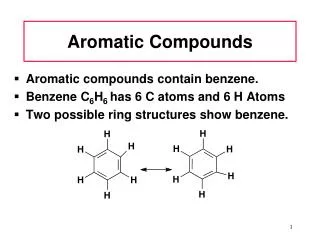
Aromatic ring (benzene ring) • benzene is perfectly hexagonal—all six carbon-carbon bonds have the same length, intermediate between that of a single and that of a double bond.
Why aromatic compound is “aromatic”? • Many molecules containing benzene rings are quite fragrant and pleasant smelling.
Resonance structure • The ring-like structure of benzene can be written in either of two Resonance Structure. • Definition of resonance structure: consists of alternating single and double bonds between carbon atoms. • The actual arrangement of electrons in the carbon is a mixture of both resonance structures. • Benzene is frequently represented as follows:
Why Benzene’s resonance structures are unusually stable? • when resonance is involved, the real structure is more stable than we would expect from any of the structures we write using the one line
Practice time Page 232 / # 29 (b)
How to name the Aromatic Structures? • General formula: ____benzene - attached F, Cl, Br, and I aroms are called “fluoro”, “chloro”, “bromo”, “iodo”. These are called halo group - if more than one of the same kind of halogen is present, use the prefixes di, tri, etc - if a compound contains both alkyl and halo groups, list the attached groups in alphabetical order. EXAMPLES~~
Practice Time • Page 232 / # 31 (a) (e)
Practice Time • Page 232 / # 30 (a) (c) (e)
Today’s homework • Page 232 / # 30 b d f # 31 b d f
Thank You!!! • If you have any questions, please ask us through the wikispaces! We will respond as soon as possible!
Reference • Hebden Chemistry 11 Second printing, October 1998. Written by James A. Hebden, Ph.D • http://en.wikipedia.org/wiki/Aromatic_compounds May 25, 2010