The RENAAL Sub-study
The RENAAL Sub-study. Reduction of end points in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL) sub-study. Reference Brenner BM, Cooper ME, et al . Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL) sub-study. Diabetes Care . 2004;27:874–879.

The RENAAL Sub-study
E N D
Presentation Transcript
The RENAAL Sub-study Reduction of end points in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL) sub-study Reference Brenner BM, Cooper ME, et al. Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL) sub-study. Diabetes Care. 2004;27:874–879.
Background Diabetes is highly prevalent in Asia and it is predicted to have the largest population of patients with diabetes who are at high risk for renal disease. In the Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL) study, approximately 17% of 1,513 type 2 diabetic patients with clinical proteinuria were Asians.
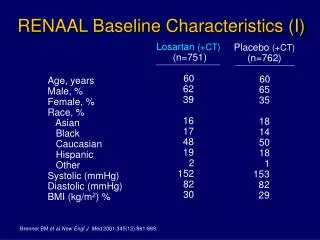
Method • Study design: The RENAAL sub-study is a double-blind, randomized, placebo-controlled study. • Study population: 252 Asian patients (aged 31–70 years) with type 2 diabetic nephropathy and serum creatinine values between 1.3 and 3.0 mg/dl, with a lower limit of 1.5 mg/dl for male patients weighing >60 kg. • Treatment regimen: Patients were randomly assigned to receive either Losartan 50 mg or placebo once a day along with conventional anti-hypertensive therapy. The dose was increased after 4 weeks to 100 mg once a day if the trough BP of the patient was below target. • End point: The primary end point was the time taken for the first event of a doubling of serum creatinine concentration, end-stage renal disease or death. The secondary end point was morbidity and mortality from cardiovascular causes like myocardial infarction, fatal or non-fatal stroke and first hospitalization from heart failure.
Result Losartan significantly reduced the risk for the primary composite end point in the RENAAL Asian population by 35% as compared to placebo. The risk of renal outcomes of ESRD or DsCr was 36% lower with losartan compared to placebo.
The risk of renal outcomes of ESRD or DsCr was 36% lower with losartan compared to placebo. The rate of decrease in renal function was reduced by 30.7% with losartan as compared to placebo.
Conclusion The risk of developing the primary end point of double creatinine levels and ESRD and all cause mortality was significantly reduced with losartan treatment in the Asian patients. Losartan was found to reduce proteinuria and progression of renal disease and was also well tolerated. In the Asian population of the RENAAL study, losartan was found to reduce proteinuria and progression of renal disease and was also well tolerated.