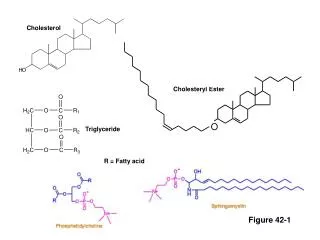
Cholesterol
Cholesterol. Amy M. Sharma PHM 142 REM University of Toronto Sept. 27 2012. HDL. Stage 4 GOOD HDL : Reverse cholesterol transport and function

Cholesterol
E N D
Presentation Transcript
Cholesterol Amy M. Sharma PHM 142 REM University of TorontoSept. 27 2012
HDL • Stage 4 GOOD HDL : Reverse cholesterol transport and function • 1. HDL is synthesized and secreted from the liver and the intestine. HDL contains 65% protein + free fatty acids, cholesterol, triacylglyceride and phospholipids. • 2 Function: • HDL picks up cholesterol released into the plasma from dying cells • and from membranes undergoing turnover and returns it to the liver • 3. HDL contains cholesterol, cholesterol ester, phospholipid and Lecithin:Cholesterol Acyl Transferase (LCAT) - synthesised in the liver that catalyses : • LECITHIN + CHOLESTEROL • LYSOLECITHIN + CHOLESTEROL ESTER • LCAT is activated by apo-A1 and deficiency in LCAT means that HDL can’t take up cholesterol from tissue, therefore cholesterol and lecithin in tissue
Increasing HDL to decrease tissue Clt • Anacetrapib (Merck drug) NEW • Niacin best • Fibrate drugs • bile acid binding resins • Exercise , -3 fatty acids,redwine,orangejuice,beans, soy,oat bran • tr.fatty acids, high carbohydrate. decr. HDL
Cholesterol Synthesis • Essential molecule in many animals including humans • However not required from diet – all cells can synthesize it • Majority produced in the liver • Synthesis occurs in 4 stages: • 1. Condensation of 3 acetate units to form 6-carbon intd, Mevalonate • 2. Mevalonate to activated isoprene units • 3. Polymerization of six 5-carbon isoprene units to form Squalene • 4. Squalene cyclization to form four rings of steroid nucleus followed by more changes to form final cholesterol
Cholesterol Synthesis: Step 1: Mitochondria Committed and rate- limiting step: Reduction of HMG-CoA to Mevalonate
Cholesterol Synthesis: Step 2: Synthesis of isopentenyl pyrophosphate from mevalonate occurs in the PEROXISOMES
Cholesterol Synthesis: Step 3: Synthesis of squalene occurs in the peroxisomes then theendoplasmic reticulum peroxisome peroxisome e.r. Squalene synthase DIMERIZATION CoQ, heme A Dolichol-PP Prenylated proteins
Squalene Cholesterol Synthesis: Step 4: Synthesis of cholesterol occurs in the ER er P450, O2, NADPH Squalene epoxide cyclase Lanosterol diet sunlight Dehydrocholesterol Vitamin D3 NADPH P450 reductase Unsat. FA acyl CoA +cholesterol acyltransferase (ACAT) CHOLESTEROL Cholesterol ester
Drug therapy to decrease plasma Clt Statins (HMG-CoA reductase inhibitors) • Inhibit cholesterol biosynthesis in liver to decrease plasma LDL cholesterol and cut the risk of heart attacks and strokes by at least 33% • HMG CoA reductase inhibitors can induce rhabdomyolysis (test for muscle/kidney damage) • Lipitor, Zocor, Crestor lower risk of heart attack, death and stroke Mevinolin 3-hydroxy-3-methyl- glutaryl CoA Mevinolin (Fungal), a competitive inhibitor of HMG CoA reductase, resembles 3-hydroxy-3-methyl-glutaryl CoA, the substrate.
But Statins may also decrease plasma ubiquinone antioxidant HMG-CoA Isopentenyl-PP TYROSINE Dimethylallyl-PP Peroxisome 4-OH-benzoate Geranyl-PP ER, Golgi Decaprenyl- 4-OH benzoate transf. Polyprenyl-PP Farnesyl-PP Decaprenyl-PP trans-prenyltransf. Squalene synthase Dolichol N-glycosylates secretory proteins = Export glycoproteins Decaprenyl-4-OH-benzoate Squalene cholesterol LDL receptors (induced) LDL uptake LDL risk of atherosclerosis COENZYME Q Cholesterol Free Rad. Biol. Med. 29, 285-94 (2000) Lancet 356, 391-5 (2000)
Drug therapy to decrease plasma Clt Resins (hydrocarbon secretion) Bind bile acids to lower cholesterol • Cholesterol is reabsorbed from intestine by forming complexes with bile acids. Liver then replaces bile acids by oxidizing cholesterol (catalysed by CYP7A). Prescription therapeutic resins bind bile acids and prevent cholesterol reabsorption: problem of constipation, ↓absorption of fat sol. vitamin A,D,E,K • e.g. colestipol • cholestyramine • colesevelam Nonprescription bulk forming laxatives (soluble fibres) • Psyllium husks (metamucil) • Ispaghula husks • Oat bran (-glucan binds bile acids ) • Action of bile acid binders • cholesterol excretion • hepatic cholesterol 7a hydroxylase (CYP7A) activity which oxidizes cholesterol to bile acids. (feedback inhibitor is normally bile acids)
Drug therapy to decrease plasma Clt Hypolipidemicieantihyperlipidemicfibrate drugs • CLOFIBRATE: 2g/day • (also: Gemfibrozil) 1. ↑ lipoprotein lipase activity 2. ↑ fatty acid oxidation by inducing PEROXISOMES serum triglycerides serum triglyceride-rich lipoprotein 3.Antioxidant action prevent LDL oxidation
Perioxisomes • Peroxisomes(microbodies) • found in virtually all eukaryotic cells. • Involved in the catabolism of very long chain fatty acids, branched chain fatty acids, D-amino acids, polyamines, and biosynthesis of plasmalogens, i.e. ether phospholipids critical for the normal function of mammalian brains and lungs. • Major function: breakdown of very long chain fatty acids through beta-oxidation. • converted to medium chain fatty acids to mitochondriacarbondioxide and water.
PEROXISOME (numerous genetic diseases) Peroxisomal fatty acid b-oxidation forms H2O2 which is removed by catalase located in the peroxisomes. Medium-chain fatty acids (C8-18) prefer mitochondrial b-oxidation that doesn’t form H2O2. Long chain or 3 or branched fatty acids MITOCHONDRIA b-oxidation Acyl carnitine Synthase Acetyl CoA oxidase* Shorter-chain fatty acid Fatty acyl CoA NADH + Acetyl CoA O2 H2O2 heat catalase Cholesterol CoQ10* Bile acids H2O + O2 H2O2 also formed by peroxisomal glycolate/glyoxylate oxidases, xanthine oxidase, uricase * Peroxisomes induced by peroxisome proliferators via a cytosolic receptor (PPAR) e.g., hypolipidemic drugs, e.g., clofibrate; plasticizers, e.g., phthalate (DEHP); endogenous steroids formed by the adrenal glands e.g., dehydroepiandrosterone. Ann Rev Biochem. 61, 157-97 (1992) Ann Rev Nutr. 14, 343-70 (1994)
Drug therapy to decrease plasma Clt Niacin(Vitamin B3) • Deficiency causes pellagra (rough photo-sensitive skin, dementia, etc) Flour now fortified with niacin; B 1 thiamine; B2 riboflavin tryptophan niacin nicotinamide NAD NADP NADPH derivatives NAD, NADH, NAD+, and NADP play essential roles in energy metabolism and DNA repair. • Niacin 1.5-3g/day ↓ plasma LDL cholesterol & triglycerides; best for ↑ HDL But early hot flashes so use slow delivery formula Rare hepatotoxicity or hyperglycemia
Drug therapy to decrease plasma Clt Blocking intestinal cholesterol permease Ezetimibe- drug that blocks cholesterol uptake by inhibiting intestinal sterol permease (packaged with a statin).
Natural therapy to decrease plasma Clt Plant sterols/stanols Ezetimibe- drug that blocks cholesterol uptake by inhibiting intestinal sterol permease (packaged with a statin). • Sitosterol , Sitostanol • Clt lowering action of plant sterols on the diet Plant sterol not absorbed by gut (2g/day) so inhibits gut absorption of cholesterol from diet. “functional margarine”) e.g. Becel pro-activ.in Loblaws
Natural therapy to decrease plasma Clt • Chitosan (shellfish exoskeleton) (LIBRACOL is polychitosamine: amine groups bind cholesterol) • Policosanol (sugar cane wax or rice wax alcohol ie. Octacosanol) CH3(CH2)26CH2OH)
Summary: • DIETARY WAYS OF DECREASING THE ATHEROSCLEROSIS RISK: • cholesterol and saturated fatty acids plant stanols (2g/margarine day) • polyunsaturated fatty acids • which cholesterol oxidation to bile acids • LDL catabolism • cholesterol excretion into intestine • 3. smoking, obesity, lack of exercise, low Ca2+ • high HDL in premenopausal women protects • but not after menopause.
Summary:Dietary mechanisms to decrease cholesterol are additive (e.g., use in patients resistant or intolerant to statins). • Decrease intestinal bile acids by binding them to viscous fibres, e.g., oats (b-glucan), barley, psyllium (metamucil), egg plant,ochra. Glucan is also a soluble fibre & an antioxidant which prev. oxidn of PUFA & cholesterol. Amer.J.Clin.Nutrition 75(2002)834-9. 2. Competitive inhibition of cholesterol absorption from the gut, e.g., plant sterols margarine, almonds, flaxseed. 3. Increase LDL receptor-mediated LDL cholesterol uptake and degradation, e.g., soy proteins, soy milk. 4. Decrease oxidized LDL using antioxidants, e.g., almonds (Vit E), soy proteins (isoflavones).
Bile: function & importance Bile (gall) - fluid produced by the liver of most vertebrates • Composed of water, bile salts, mucus and pigments, fats, inorganic salts and cholesterol • Main functions: • Essential in the process of digestion of lipids from the small intestine • For protection of small intestines from oxidative damage • For excretion of endogenous xenobiotic compounds Also: • Aids in absorption of fat-soluble vitamins A, D, E, K • Bile salts are also bactericidal (protect from microbial products in foodstuff)
Bile: dysfunction & implications • Drugs that slow/block bile flow from the liver to the gallbladder & gut may cause liver failure. e.g. chlorpromazine,prochlorperazine, penicillin ampicillin,estradiol, nitrofurantoin, sulindac. • Symptoms include dark urine, pale stool, jaundice, fever/rash persistent itching. MUST discontinue the drug.
Bile: potential for toxicity • Bile acids are cytotoxic to hepatocytes (can cause liver failure via free radical formation). • Mechanism: • Reduces Fe3+ which reduces H2O2 to form hydroxyl radicals and reactive oxygen species (ROS) • Radicals oxidize nucleic acids, proteins, and unsaturated lipids to form other radicals. • BUT… • Antioxidants & blockers of mitochondrial permeability transition prevent apoptosis
Cholestasis defined Cholestasis - any condition in which the flow of bile from the liver is blocked. i.e. where bile cannot flow from the liver to duodenum • Obstructive cholestasis - mechanical blockage in the duct system such i.e. as a results of gallstone or malignancy • Metabolic cholestasis - disturbance in bile formation due to genetic defects or as an adverse effect of a drug
Extra-hepatic cholestasisCholestasis occurring outside the liver • Caused by blockage of bile duct or ducts • Possible causes: - bile duct tumors - Pancreatic tumor or pseudocyst - Cysts - Narrowing of the bile duct - Stones - Pancreatitis - Pressure on an organ due to tumor or nearby mass
Intra-hepatic cholestasisCholestasis occurring within the liver • Caused by significant blockage of small ducts or by disorders, such as hepatitis, that impair the body's ability to eliminate bile • Possible causes: - Alcoholic liver disease - Amyloidosis - Bacterial abscess • Lymphoma • Drugs - Primary biliary cirrhosis - Tuberculosis - Sepsis, - Viral hepatitis
G) BILE ACID SYNTHESIS BY LIVER Endoplasmic reticulum (except CYP27) Hepatocyte death Biochemistry. 31, 4737-49, (1992)
Ga) BILE ACID SYNTHESIS BY LIVER (cont) Then efflux into bile and stored in gall bladder. Then released by bile duct into upper-small intestine (ileum). Then metabolised (deconjugation (CO2), dehydroxylation) by anaerobic bacteria of colon to deoxycholate, lithocholate, urodeoxycholate. Then actively reabsorbed and recirculates via liver 8 times / day.