Coordination Complexes and Transition Metals in Action
760 likes | 2.18k Vues
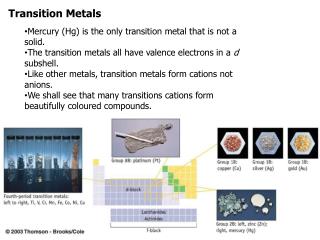
Coordination Complexes and Transition Metals in Action. Al 2 O 3 crystal with traces of Cr 3+ (ruby). Spring and summer – chlorophyll and xanthophyll. Fall – xanthophyll colors dominate. Plants and Animals. chlorophyll. heme. Colors of Chromium. Cr(NO 3 ) 3. CrCl 3. K 2 CrO 4.

Coordination Complexes and Transition Metals in Action
E N D
Presentation Transcript
Coordination Complexes and Transition Metals in Action Al2O3 crystal with traces of Cr3+ (ruby) Spring and summer – chlorophyll and xanthophyll Fall – xanthophyll colors dominate
Plants and Animals chlorophyll heme
Colors of Chromium Cr(NO3)3 CrCl3 K2CrO4 K2Cr2O7 Cr3+ Cr6+
Coordination Compound and Complex Coordination Compound is [Co(NH3)6]Cl3 [Co(NH3)6]Cl3 [Co(NH3)6]3+ + 3 Cl- Coordination Complex is [Co(NH3)6]3+
Metal ion – usually transition metals with empty valence orbitals Specifically empty d orbitals Act as Lewis acid (electron pair acceptor) Ligand – complexing agent bound to (surrounding) the metal ion (Lewis base) Normally ligands are anions or polar molecules Anion (CN-) Polar molecule (NH3) Donor atom Components of Complex (Coordination Sphere) [Co(NH3)6]3+
Metal ion Oxidation number Co = ? Ligand Charge on ligand NH3 = ? Characteristics of Complex (Coordination Sphere) Charge of Complex – sum of charges on the central metal ion and the surrounding ligands What is the charge of the complex? [Co(NH3)6]Cl3 Coordination number – The number of donor atoms attached to the metal.
Example Problem • Indicate the coordination number of the metal and the oxidation number of the metal in each of the following complexes: Na2[CdCl4] [Co(NH3)4Cl2]Cl K2[MoOCl4] [Zn(en)2]Br2
Types of Ligands • Monodentate ligand NH3, H2O, Cl- • Bidentate ligand ethylenediamine (en) • Polydentate ligand ethylenediaminetetraacetate ion (EDTA)4-
Chelating Agent • Polydentate ligands (including bidentate) are called chelating agents because they appear to grasp the metal between donor atoms
Example Ligands What kind of ligands are these examples?
Chelate Effect • Chelating agents form more stable complexes with metal ions than monodentate ligands Ni2+(aq) + 6NH3(aq) [Ni(NH3)6]2+(aq) Kf = 4 x 108 Ni2+(aq) + 3 en (aq) [Ni(en)3]2+(aq) Kf = 2 x 1018 Sequestering agents – because the chelating agents can be used to remove or separate ions removal of ions from hard water removal of trace metals from food removal of heavy metal ions from blood
Geometry of Complex • Common coordination # of 4 square planar Transition metal ions with 8 d electrons Tetrahedral Most common
Geometry of Complex • Coordination # 6 Octahedral
Effect of Ligand on Coordination Number • The larger the size of the ligand, the fewer ligands that can get close enough to bind to the central metal. [FeF6]3- [FeCl4]- • Ligands which provide negative charge to the complex reduce the coordination number. [Ni(NH3)6]2+ [Ni(CN)4]2-
Metal Complexes • Distinct chemical properties different from the metals and ligands from which they were formed. • Different colors • Different electrochemical properties • Different solubility properties
Nomenclature • In naming salts, the name of the cation is given before the name of the anion. [Mo(NH3)3Br3]NO3 Cation Anion = nitrate
Nomenclature • Within a complex ion or molecule the ligands are named before the metal. Ligands are listed in alphabetical order, regardless of charge on the ligand. Prefixes that give the number of ligands are not considered part of the ligand name in determining alphabetical order. [Mo(NH3)3Br3]NO3 Ammonia, bromide, molybdenum
Nomenclature • The names of the anionic ligands end in the letter o, whereas neutral ones ordinarily bear the name of the molecules. [Mo(NH3)3Br3]NO3 Example ligand names NH3 – ammine CO – carbonyl NO - nitrosyl H2O – aqua CN- – cyano en - ethylenediammine Ammine, bromo, molybdenum
Nomenclature • Greek prefixes (di, tri, tetra, penta, hexa) are used to indicate the number of each kind of ligand when more than one is present. [Mo(NH3)3Br3]NO3 If the ligand itself contains a prefix, then these prefixes are used for the ligand name (bis-, tris-, tetrakis-, pentakis-, etc.). [Ru(bipy)3]Cl3 triamminetribromomolybdenum tris-bipyridineruthenium
Nomenclature • If the complex is an anion, its name ends in –ate. K3[V(C2O4)3] cation = potassium anion = trioxalatevanadate
Nomenclature • The oxidation number of the metal is given in parentheses in Roman numerals following the name of the metal. [Mo(NH3)3Br3]NO3 [Ru(bipy)3]Cl3 K3[V(C2O4)3] triamminetribromomolybdenum(IV) nitrate tris-bipyridineruthenium(III) chloride Potassium trioxalatevanadate(III)
Isomers Hydrate isomer Ionization isomer
Structural Isomerismhave the same numbers and kinds of atoms, but differ in the bonds that are present. Ionization isomer – exchange of ligand with an anion or neutral molecule [CoBr(NH3)5]SO4 and [CoSO4(NH3)5] Br Hydrate isomer – the exchange of H2O molecule with another ligand [CrBr(H2O)6]Cl3 and [CrClBr(H2O)5]Cl2*H2O Coordination isomers- differ due to the exchange of one or more ligands between a cationic complex and an anionic complex [Cr(NH3)6][Fe(CN)6] [Fe(NH3)6][Cr(CN)6] Linkage isomers- contain the same ligand coordinated to the metal through different donor atoms[Pd(NH3)3SCN]+ and [Pd(NH3)3NCS]+
Linkage Isomerism Nitro Nitrito
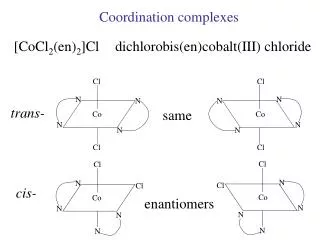
Stereoisomerism Geometric isomers have the same number and kinds of bonds, but differ in the relative positions of the ligands. Optical isomers rotate the plane of polarized light in opposite directions.
Optical Isomers Rotate the plane of polarized light in opposite directions. Levrorotatory – rotate left Dextrorotatory – rotate right
Optical Isomers Chiral molecules have mirror-image structures that cannot be superimposed. Only chiral molecules are optically active. Enantiomers are chiral molecules of each other. Racemic Mixture occurs when equal amounts of each enantiomer are mixed. When this happens, the optical activity of each is canceled by the other.
Properties of Coordination Complexes • Color– Many coordination complexes exhibit a wide variety of colors, that depend on the metal, its oxidation state, and the ligands present. • The observed colors result from the absorption of light in the visible region by the complexes. Color Exhibited Colorless Partially filled d orbital Totally filled or empty d orbital (d0 and d10)
Color Exhibited [Cr(NH3)6]3+ [Fe(SO4)(H2O)4] Colorless [Cd(NH3)4](NO3)2 Na[AlCl4] Color Exhibited or Colorless
Crystal Field Theory Crystal field theory assumes electrostatic interactions between the negative or neutral ligands and the positive metal ion lower the energy of the system. • Anionic ligands – electrostatic attraction • Neutral ligands – ion dipole
Lowered Energy of Metal/Ligand Complex Crystal Field – repulsive interaction between electrons in the ligands and the d orbital electrons in the metal
Consequence:d electron repulsion Crystal Field • The negative ligands repel the electrons in the metal ion d orbitals. • The repulsion energy of d electrons depends on the orientation of the orbital, relative to the location of the negative ligands.
d orbital splitting D = Energy gap – the energy necessary for an electron to move across the gap is similar to energy of a visible light photon Explains why d0 or d10 transition elements do not show color
Electron Configurations Co3+ a d6 ion
Electron Configurations • High spin • Low spin Weak Field ligand Spin pairing energy is the energy required to pair 2 electrons in orbital Strong Field ligand
Properties of Coordination Complexes • Paramagnetism- a property due to unpaired electrons, is common among transition metal complexes. • Different complexes of the same metal ion, may have different numbers of unpaired electrons. Predict the magnetic properties of: [Fe(H2O)6]2+ [Fe(CN)6]4-
Energy Calculation • The complex [Ti(H2O)6]3+ absorbs light of wavelength 510 nm. What is the crystal field d orbital splitting energy (D) for the complex?
Metal and Oxidation # • Color– Many coordination complexes exhibit a wide variety of colors, that depend on the metal, its oxidation state, and the ligands present. [Cr(H2O)6]3+ [V(H2O)6]2+ The larger the charge on the metal ion involved in the complex, the more metal-ligand interaction. Therefore, D will be larger when the oxidation state of the metal is larger.
Increased Interaction Between Metal and Ligand: Increased Crystal Field Crystal Field – repulsive interaction between electrons in the ligands and the d orbital electrons in the metal
Crystal Field Theory Tetrahedral Tetrahedral complex always have high spin because D is small