Protein Purification
Protein Purification. Protein Purification. What do you know about proteins? Why do we need to purify proteins? What are you curious about?. Bacterial chromosome. What is Transformation?. Uptake of foreign DNA, often a circular plasmid. Plasmid.

Protein Purification
E N D
Presentation Transcript
Protein Purification • What do you know about proteins? • Why do we need to purify proteins? • What are you curious about?
Bacterial chromosome What is Transformation? Uptake of foreign DNA, often a circular plasmid Plasmid Allow bacteria to grow for 1-3 days on plate with ampicillin. Bacterial chromosome Bacteria now express cloned fluorescent protein…
Through transformation, bacteria now express cloned fluorescent protein.
Purified Fluorescent Proteins Roger Tsien and Osamu Shimomura Tsien Osamu Shimomura
What is Protein Purification? Purify a single protein of interest from over 4,000 naturally occurring E. coli gene products.
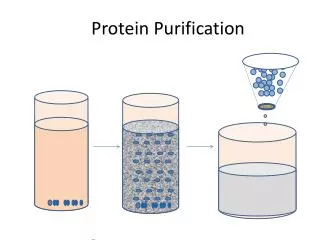
Purification Steps Supernatant Snap freeze on dry ice Pellet 3.Mix supernatant with nickel beads • Lyse (cut) open the cells 2. Centrifuge to create pellet
Column Chromatography 4. Pass the supernatant through the column 5. Add elution buffer 6. End with a pure sample containing only the fluorescent protein
The His tag us how the protein attaches to the Nickel bead his-his-his-his-his-his Fluorescent Protein with “his tag” The “his tag”
Purpose of the Nickel Beads • Nickel bead binds to His tag of FP (like two magnets) • Now the Fluorescent Proteins are attached to the nickel beads and will not be able to flow through the column with the other proteins. Ni2+
Separating FP from other proteins Fluorescent Proteins The Nickel beads are too big to pass through the column, so the FP’s that are stuck to nickel beads are not able to flow through the cotton. All other proteins will flow through cotton ball into waste tube
Elution Imidazole • FP are separated from nickel beads by the imidizole (elution buffer) • Now that FP is no longer attached to the Ni bead, it can pass through the column Ni2+ Histidine
LYSOZYME In our saliva, tears, spleen, lung, kidney High concentration in chicken egg-white (our source of lysozyme). Lysozyme was discovered accidentally in 1922 by Alexander Fleming by accident. Nasal drippings in the petri dish with bacterial culture, killing the bacterial cells. Viruses uselysozymeto break into the host bacterial cell allowing it to inject its DNA.
“Snap Freeze” Cell Lysis • This technique involves freezing and then thawing the material. • Causes cells to swell and ultimately break as ice crystals form during the freezing process and then contract during thawing. • .
The His tag us how the protein attaches to the Nickel bead his-his-his-his-his-his Fluorescent Protein with “his tag” The “his tag”
Purpose of the Nickel Beads • Nickel bead binds to His tag of FP (like two magnets) • Now the Fluorescent Proteins are attached to the nickel beads and will not be able to flow through the column with the other proteins. Ni2+
Elution Imidazole • FP are separated from nickel beads by the imidizole (elution buffer) • Now that FP is no longer attached to the Ni bead, it can pass through the column Ni2+ Histidine
Fluorescent Protein Purification From organism… …to purified protein product
Central Dogma of Molecular Biology Transcription Translation DNA---> mRNA---> Protein---> Trait
Fluorescent Proteins Green (GFP) Red (RFP)
Why Purify Proteins? In RESEARCH: Characterization of the function, structure and interactions • In MEDICINE: • Vaccines created from recombinant proteins Example: X-Ray Crystallography Example: Insulin
Engineered Fluorescent Proteins From GFP:From RFP: 1. Green 4. Cherry 2. Blue 5. Tangerine 3. Grape 6. Yellow
Protein Purification Now that you’ve purified your floursecent proteins – how else to you see that this process could be used?