Atomic Physics and Lasers
Atomic Physics and Lasers. The idea of a photon Black body radiation Photoelectric Effect The structure of the atom How does a Laser work? Interaction of lasers with matter Laser safety Applications Spectroscopy, detection of art forgery, flow cytometry, eye surgery.

Atomic Physics and Lasers
E N D
Presentation Transcript
Atomic Physics and Lasers • The idea of a photon • Black body radiation • Photoelectric Effect • The structure of the atom • How does a Laser work? Interaction of lasers with matter • Laser safety • Applications • Spectroscopy, detection of art forgery, flow cytometry, eye surgery.
The idea of a photon • What is light? • A wave? • Well yes, but…. • The wave picture failed to explain physical phenomena including : • the spectrum of a blackbody • the photoelectric effect • line spectra emitted by atoms
Light from a hot object... Vibrational motion of particles produces light(we call the light “Thermal Radiation”)
The first clue that something was very, very wrong…Blackbody radiation • What is a blackbody? • An object which emits or absorbs all the radiation incident on it. • Typical black bodies • A light globe • A box with a small hole in it.
Example of a Blackbody A BLACKBODY
Example of a Blackbody We measure radiation as a function of frequency (wavelength)
A Thermal Spectrum How does a thermal spectrum change when you change T?
Thermal Radiation T= Temp. in Kelvin Total energy emitted by an object (or Luminosity W/m2) Wavelength where flux is a maximum s = 5.7 x 10-8 W/(m2.K4) k = 2.898 x 10-3 m.K Stefan’s Law Wien’s Law
Matter at some temperature T Oscillators Light and matter interact • The spectra we have looked at are for ideal objects that are perfect absorbers and emitters of light Light is later emitted Light is perfectly absorbed A BLACKBODY
Not so good here F l u x Okay here l F l u x Problems with wave theory of light • Take a Blackbody witha temperature, T • Calculate how the spectrum would look if light behaved like a wave (Lord Rayleigh) • Compare with what isactually observed
Max Plank Solved the problem in 1900 • Oscillators cannot have any energy! They can be in states with fixed amounts of energy. • The oscillators change state by emitting/absorbingpackets with a fixed amounts of energy Max Plank
Atomic Physics/Blackbody • Max Planck (1858-1947) was impressed by the fact • spectrum of a black body was a universal property. E =nhf • To get agreement between the experiment and the theory, Planck proposed a radical idea: Light comes in packets of energy called photons, and the energy is given by E= nhf The birth of the quantum theory = Planck’s hypothesis
The birth of the Photon • In 1906, Einstein proved that Planck’s radiation law could be derived only if the energy of each oscillator is quantized. En = nhf ; n = 0, 1, 2, 3, 4,... h=Planck’s constant= 6.626x10 -34 J.s f=frequency in Hz; E=energy in Joules (J). • Einstein introduced the idea that radiation • equals a collection of discrete energy quanta. • G.N. Lewis in 1926 named quanta “Photons”.
Atomic Physics/Photon • The energy of each photon: E = hf h=Planck’s constant f=frequency Ex. 1. Yellow light has a frequency of 6.0 x 1014 Hz. Determine the energy carried by a quantum of this light. If the energy flux of sunlight reaching the earth’s surface is 1000 Watts per square meter, find the number of photons in sunlight that reach the earth’s surface per square meter per second. Ans. 2.5 eV and 2.5 x 10 21 photons / m 2 /s
Shining light onto metals Light in Nothing happens METAL
Different Energy Light in electrons come out Shining light onto metals METAL
The Photoelectric Effect • When light is incident on certain metallic surfaces, electrons are emitted = the Photoelectric Effect(Serway and Jewett 28.2) • Einstein: A single photon gives up all its energy to a single electron EPhoton =EFree + EKinetic Need at least this much energy to free the electron Whatever is left makes it move
Kinetic Energy of electron Different metals Frequency of Light fo Threshold frequency The Photoelectric Effect
Metal plate Application of Photoelectric Effect Soundtrack on Celluloid film To speaker
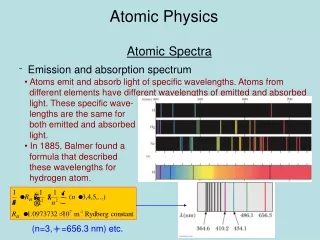
Another Blow for classical physics: Line Spectra • The emission spectrum from a rarefied gas through which an electrical discharge passes consists of sharp spectral lines. • Each atom has its own characteristic spectrum. • Hydrogen has four spectral lines in the visible region and many UV and IR lines not visible to the human eye. • The wave picture failed to explain these lines.
Atomic Physics/Line spectra 400 500 600 (nm) H Emission spectrum for hydrogen The absorption spectrum for hydrogen; dark absorption lines occur at the same wavelengths as emission lines.
-0.85 n=4 -1.51 n=3 Paschen IR n=2 -3.39 Balmer Visible R =Rydberg Constant = 1.09737x10 7m-1 -13.6 1 1 1 R ( nm2 n=1 Lyman UV ) Atomic Physics/Line Spectra
So what is light? • Both a wave and a particle. It can be both, but in any experiment only its wave or its particle nature is manifested. (Go figure!)
Two revolutions: The Nature of light and the nature of matter • Light has both a particle and wave nature: • Wave nature: • Diffraction, interference • Particle nature • Black body radiation, photoelectric effect, line spectra • Need to revise the nature of matter (it turns out that matter also has both a particle and wave nature
The spectrum from a blackbody • Empirically: • (max)T = constant, • Hotter = whiter • The wave picture (Rayleigh-Jeans) failed to explain the distribution of the energy versus wavelength. UV Catastrophe!!!! 6000K Rayleigh- Jeans Relative Intensity Observed 5000K 0 2 4 6 8 10 (10 -7 m)
Photoelectric Effect Light in e Electron out METAL
The Photoelectric Effect • Photoelectric effect=When light is incident on certain metallic surfaces, photoelectrons are emitted. • Einstein applied the idea of light quanta: In a photoemission process, a single photon gives up all its energy to a single electron. Energy of photon Energy to free electron = + KE of emitted electron
Atomic Physics/Photoelectric Effect =work function; minimum energy needed to extract an electron. hf = KE + KE x fo = threshold freq below which no photoemission occurs. x x x f0 f, Hz
Atomic Physics/The Photoelectric Effect-Application The sound on a movie film Sound Track Phototube Light Source speaker
The photoelectric effect • Photoelectric effect=When light is incident on certain metallic surfaces, photoelectrons are emitted. • Einstein applied the idea of light quanta: In a photoemission process, a single photon gives up all its energy to a single electron. Energy to free electron Energy of photon KE of emitted electron = +
The Photoelectric Effect experiment Metal surfaces in a vacuum eject electrons when irradiated by UV light.
PE effect:5 Experimental observations • If V is kept constant, the photoelectric current ip increases with increasing UV intensity. • Photoelectrons are emitted less than 1 nS after surface illumination • For a given surface material, electrons are emitted only if the incident radiation is at or above a certain frequency, independent of intensity. • The maximum kinetic energy, Kmax, of thephotoelectrons is independent of the light intensity I. • The maximum kinetic energy, Kmax of the photoelectrons depends on the frequency of the incident radiation.
Failure of Classcial Theory Observation 1: is in perfect agreement with classical expectations Observation 2: Cannot explain this. Very weak intensity should take longer to accumulate energy to eject electrons Observation 3: Cannot explain this either. Classically no relation between frequency and energy. Observations 4 and 5: Cannot be explained at all by classical E/M waves. . Bottom line: Classical explanation fails badly.
Quantum Explanation. • Einstein expanded Planck’s hypothesis and applied it directly to EM radiation • EM radiation consists of bundles of energy (photons) • These photons have energy E = hf • If an electron absorbs a photon of energy E = hf in order to escape the surface it uses up energy φ,called the work function of the metal • φis the binding energy of the electron to the surface • This satisfies all 5 experimental observations .
Photoelectric effect • hf = KE + φ • ( φ=work function; minimum energy needed to extract an electron.) • fo = threshold freq, below which no photoemission occurs KE x . x x x f0 f (Hz)
Application: Film soundtracks Sound Track Phototube Light Source speaker
5m Example: A GaN based UV detector This is a photoconductor
Choose the material for the photon energy required. • Band-Gap adjustable by adding Al from 3.4 to 6.2 eV • Band gap is direct (= efficient) • Material is robust
Characterization of Detectors • NEP= noise equivalent power = noise current (A/Hz)/Radiant sensitivity (A/W) • D = detectivity = area/NEP • IR cut-off • maximum current • maximum reverse voltage • Field of view • Junction capacitance
Photomultipliers e e e e hf e e PE effect Secondary electron emission Electron multiplication
hf e Anode Dynode -V Photomultiplier tube • Combines PE effect with electron multiplication to provide very high detection sensitivity • Can detect single photons.
Microchannel plates • The principle of the photomultiplier tube can be extended to an array of photomultipliers • This way one can obtain spatial resolution • Biggest application is in night vision goggles for military and civilian use
Microchannel plates • MCPs consist of arrays of tiny tubes • Each tube is coated with a photomultiplying film • The tubes are about 10 microns wide http://hea-www.harvard.edu/HRC/mcp/mcp.html
MCP array structure http://hea-www.harvard.edu/HRC/mcp/mcp.html
MCP fabrication http://hea-www.harvard.edu/HRC/mcp/mcp.html
Disadvantages of Photomultiplers as sensors • Need expensive and fiddly high vacuum equipment • Expensive • Fragile • Bulky
Photoconductors • As well as liberating electrons from the surface of materials, we can excite mobile electrons inside materials • The most useful class of materials to do this are semiconductors • The mobile electrons can be measured as a current proportional to the intensity of the incident radiation • Need to understand semiconductors….