Element
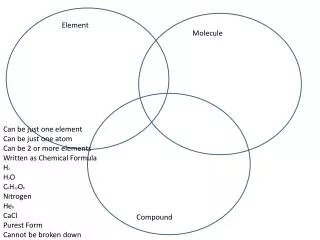
Elements and Compounds. Organic Compounds. Soaps and Detergents. Element. Compounds. Objectives:. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Preparation of soaps. Explanation of detergents.


Element
E N D
Presentation Transcript
Elements and Compounds Organic Compounds Soaps and Detergents Element Compounds Objectives: A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Preparation of soaps. Explanation of detergents. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds. Cleansing action of soaps and detergents.
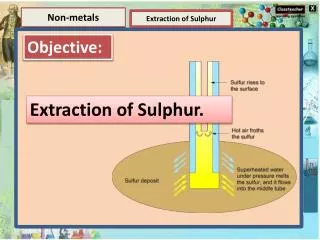
Elements and Compounds Non-metals Organic Compounds Extraction of Sulphur Soaps and Detergents Element Soaps Compounds Soaps are sodium (or potassium) salts of long-chain alkanoic acids. The ionic head of soaps is always a carboxylate group (–COO-). A common soap is sodium stearate: A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
Elements and Compounds Non-metals Organic Compounds Extraction of Sulphur Soaps and Detergents Element Preparation of Soaps Compounds Vegetable oil (ground nut oil, caster oil etc.) or animal fat when heated with sodium hydroxide (NaOH), sodium salt of fatty acid and glycerol are formed. This process of preparing soap is called saponification. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Vegetable oil + Sodium Glycerol + Sodium salt of fatty or Animal fat hydroxide acid (Soap) Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
Elements and Compounds Non-metals Organic Compounds Extraction of Sulphur Soaps and Detergents Element Detergents Compounds We use detergents every day, in one form or another. Like plastics and alkanols, most detergents are made from petroleum products. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. The chemical substance used to remove the dirt sticked to the surface and does not harm the surface of a thing is called detergent. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
Elements and Compounds Non-metals Organic Compounds Extraction of Sulphur Soaps and Detergents Element Detergents Compounds Chemically, detergents are the sodium salts of organic sulphonic acids. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. The detergent anion consists of two parts: (1) An ionic group (the ‘head’) (2) A hydrocarbon chain (the ‘tail’) Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
Elements and Compounds Non-metals Organic Compounds Extraction of Sulphur Soaps and Detergents Element Detergents Compounds The cleansing effect of the detergent is more effective because Ca2+ and Mg2+ ions present in hard water do not give precipitates with ions which are in the soluble form. They remain in the solution and so detergent in more quantity is not used. Hence, the use of detergent has increased. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds. The cleansing process of soap and detergent is same.
Elements and Compounds Non-metals Organic Compounds Extraction of Sulphur Soaps and Detergents Element Cleansing Action of Soaps Compounds A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds. Soaps and detergents are used for removing dirt from clothes.