Element
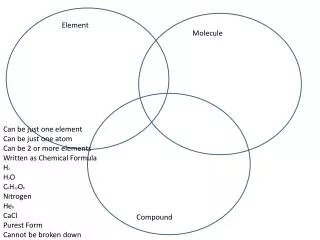
Element. A pure substance that cannot be broken down into simpler substances by physical or chemical means. Elements are considered pure …. …because all of the particles are identical. The smallest possible particle of an element is called an atom of that element.


Element
E N D
Presentation Transcript
Element A pure substance that cannot be broken down into simpler substances by physical or chemical means.
Elements are considered pure… …because all of the particles are identical. The smallest possible particle of an element is called an atom of that element.
How are elements found? • Some elements are found in nature in their pure, natural form. (Gold & copper) • Other elements are chemically and/or physically combined with other elements in nature and need to be separated from the other elements to get a pure sample.
Scientists made a list of all the known elements and organized it on a chart (table) called the Periodic Table of the Elements. Periodic Table of Elements
If it is an element, it is on the table (chart). Periodic Table of Elements If it is not an element, it is not on the table (chart).
Element Names and Symbols • Some element names match their symbol nicely. • Hydrogen (H) • Helium (He) • Oxygen (O) • Carbon (C)
Other Elements do not seem to match their symbol at all. • Gold (Au) • Silver (Ag) • Mercury (Hg) • This is because their symbol matches their name from a different language. • Gold is Aurum in Latin so the Au matches it’s Latin name. • Silver is Argentium in Latin so the Ag matches it’s Latin name. • Mercury is Hydragyrm in Latin so the Hg matches it’s Latin name.
Scientists from all over the world use the same symbols and the same Periodic Table even if they have different common names for elements. The symbol for gold is always (Au) even if its common name is: • Oro in Italian - Kin in Japanese • Or in French - Jin l in Chinese • Guld in Swedish - Geum in Korean • Zloto in Polish - Thongkam in Thai • Auksas in Lithuanian - Emas in Indonesian • Dhahab in Arabic - Kulta in Finnish • Zahaz in Hebrew - Chrysos in Greek http://elements.vanderkrogt.net/ptable.php
Atom • The smallest possible particle of an element is called an atom of that element.
Compound • A pure substance made of two or more elements that are chemically combined. • This new substance usually has very different properties than the original elements combined to form it. • Each particle of a compound is called a molecule of that compound.
Examples of Compounds • Water (H20) is made of two parts Hydrogen and one part Oxygen. • Hydrogen is a clear gas that is highly volatile and explodes if placed near a flame. • Oxygen is a clear gas that reacts with many substances and is required for most things to burn. • When chemically combined they form water which is a clear liquid that will not burn and does not react with many substances.
H2O molecules • Hydrogen (white) • Oxygen (purple) Again, it is a pure substance and all the particles are identical to each other. But, each particle is made of more than one element.
Table Salt (Sodium Chloride) NaCl • Table salt is made of one part sodium and one part chlorine. • Sodium (Na) is a silver solid that burns violently when it touches water. • Chlorine (Cl) is a yellowish gas that is so poisonous that it is illegal to use in war. • When Chemically combined they form the compound sodium chloride (NaCl) which is table salt, a white solid that is edible.
NaCl molecules • Sodium (purple) • Chlorine (green)
Mixture • When two or more compounds or elements are physically (but not chemically) combined. • The original substances keep their properties and no new substances are created • They can be separated by physical means. • They are considered “not pure substances”.
Examples of Mixtures • Sugar water - When sugar is mixed with water, the sugar and the water keep their properties because they still exist. • The mixture can be separated by physical means.
Identify each as an element, compound or mixture. Element Mixture Compound Element
Chemical Symbols • Each element has its own symbol • These symbols always begin with a capital letter • If they have a second or third letter they are always lower case C for Carbon O for Oxygen Co for Cobalt
Chemical Formulas • The symbol for a compound is composed of more than one chemical symbol. • The chemical formula for carbon dioxide is CO2. • This means that a molecule of carbondioxide is composed of one atom of carbon and two atoms of oxygen
Chemical Equations • A chemical equation is a set of symbols that describes what is occurring in a chemical reaction. • In the equation Na + Cl NaCl Na (sodium) and Cl (chlorine) are the reactants and NaCl (salt) is theproduct.
Reactants and Products • The reactants are the substances that you have originally, before the chemical change and the products are what is created during the chemical change.
Organization of the Periodic Table • The periodic table is organized into families based on their similar properties of reactivity. • Elements in the same vertical column will react similar when combined with a specific element. • Example: Sodium reacts with Chlorine to produce salt (NaCl). Lithium and Bromine will react similarly with Chlorine.