Element
Properties of Matter. Effect of Temperature & Pressure . Element. Objectives:. Effect of change in temperature on the states of matter. Effect of pressure change on the states of matter. Properties of Matter. Effect of Temperature & Pressure . Elements and Compounds. Element.

Element
E N D
Presentation Transcript
Properties of Matter Effect of Temperature & Pressure Element Objectives: Effect of change in temperature on the states of matter. Effect of pressure change on the states of matter.
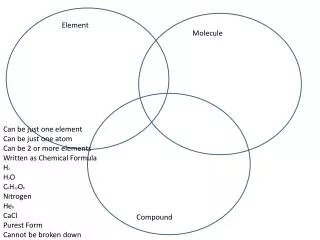
Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element States of Matter Matter exists in three different physical states − solid, liquid, and gas. These states of matter are interchangeable.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature Temperature is a major factor responsible for causing a change in the various states of matter.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature On heating a solid substance, the kinetic energy of its constituent particles increases. As a result, the particles start vibrating with greater speed. This extra energy helps the particles to overcome the inter-particle force of attraction. Soon, they leave their positions and start moving more freely. Consequently, the substance melts.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature The change of solid state into liquid state is known as fusion. The minimum temperature at which a substance starts melting and becomes a liquid at atmospheric pressure is known as its melting point.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature The temperature remains constant till all solid melts into liquid. The heat supplied is used for changing a solid state into its liquid state by overcoming the particle-particle attraction force. The solid substance absorbs heat energy without showing any rise in temperature. This heat energy is called latent heat.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature The amount of heat required to convert a unit mass of a solid into its liquid state, without a change in temperature (i.e., at its melting point), is called the latent heat of fusion. For ice, the latent heat of fusion is 334 kJkg-1.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature If heating is continued, then the kinetic energy of liquid particles increases further. This increases the velocity of the particles, and at a certain temperature, they obtain enough energy to break free from the force of attraction of the other particles. At this point, the liquid starts boiling i.e., starts changing into its gaseous state. This temperature is known as the boiling point of the liquid.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature The latent heat of vaporisation is the amount of heat required to convert a unit mass of liquid into its vapour state, without a change in its temperature. Latent heat of vaporisation of water is 2,260 kJkg-1.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature When gas is cooled beyond its boiling point, it changes to its liquid state. This process is called condensation. When a liquid is cooled beyond its freezing point, it solidifies.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Temperature When certain substances are in solid state and heat is supplied to them they do not change into liquid state but directly changes to gaseous state. This phenomenon is known as sublimation.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Pressure A change in the surrounding pressure can change the temperature that will cause a material to go from one state to another. This is because the molecules causing the pressure inhibit the molecules of the material.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Pressure When pressure is increased on liquid, it is converted to solid. This effect is less than when the pressure is increased on gaseous substance and converted to liquid. The effect of pressure on solids is negligible, more on liquids and most on gases.
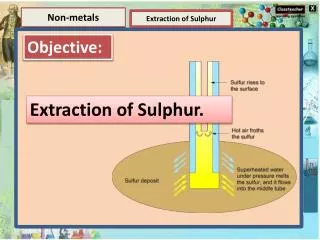
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Pressure What is dry ice and how it is obtained?
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Effect of Pressure What is dry ice and how it is obtained? If the pressure on carbon dioxide gas is increased, it is changed to solid state. Carbon dioxide is obtained as white substance in solid state whose temperature is very low. It is called dry ice. It is used to create coolness.
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Graph of Pressure versus Temperature
Elements and Compounds Properties of Matter Effect of Temperature & Pressure Elements and Compounds Element Graph of Pressure versus Temperature