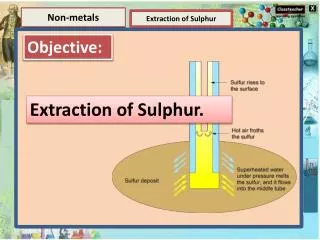
element
element. compound. Heterogeneous mixture. compound. Chemical and Physical Changes. Physical Properties. Physical properties are used to identify, describe and classify matter.


element
E N D
Presentation Transcript
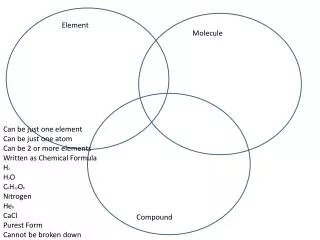
element compound Heterogeneous mixture compound
Physical Properties • Physical properties are used to identify, describe and classify matter. • Characteristic of a substance that can be observed (using your senses) without changing the substance into something else.
Chemical Properties • Chemical properties are characteristics involved when a substance interacts with another substance to change its chemical make-up.
Physical vs. Chemical Change • Physical change will change the visible appearance, without changing the composition of the material. • Boil, melt, cut, bend, split, crack • Is boiled water still water? • Chemical change - a change where a new form of matter is formed. • Rust, burn, decompose, ferment Yes, just water molecules in the gas phase
Physical changes do not result in a change of composition. • When a substance changes its composition, it undergoes a chemical change: • When pure hydrogen and pure oxygen react completely, they form pure water. In the flask containing water, there is no oxygen or hydrogen left over.
Chemical Change A change in which one or more substances are converted into different substances. Heat, light and Gases produced are evidence of a chemical change.
Compounds can only be broken down into simpler compounds or elements by chemical changes. The electrolysis of water splits the H2O molecules into H2 and O2 gas.