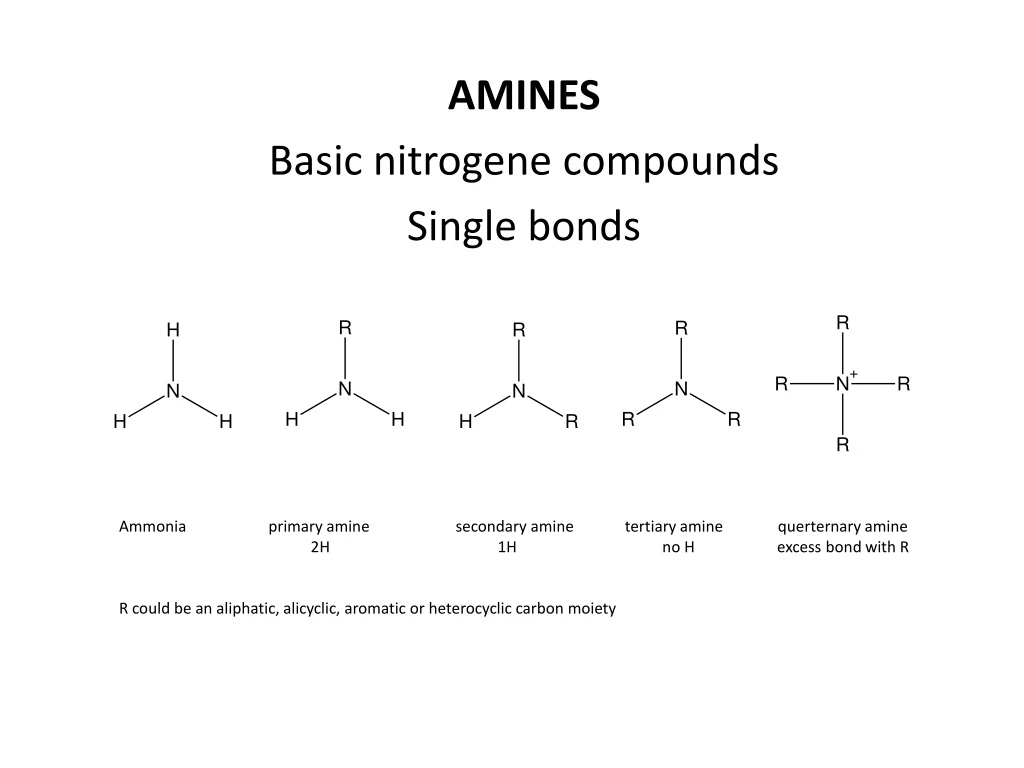
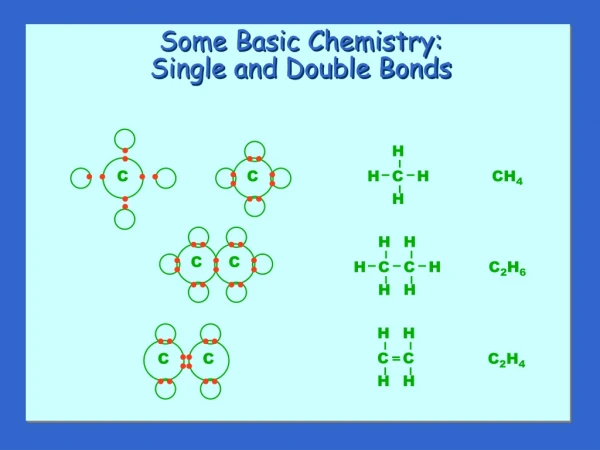
AMINES Basic nitrogene compounds Single bonds
E N D
Presentation Transcript
AMINES Basic nitrogenecompounds Singlebonds Ammoniaprimary amine secondary amine tertiary amine querternary amine 2H 1H no H excessbondwith R R could be an aliphatic, alicyclic, aromaticorheterocycliccarbonmoiety
-NH2 (amino) group a polar andbasicgroup -Aminesarehighlyreactive -Nomenclature: Simplyindicatethehydrocarbonemoietyfirstandthen amine ending: CH3-NH2methylamine C6H5-NH2 aniline N-methylethylamine (smallgroup as a substituent) N,N-dimethylethylamine N-ethyl-N-methylaniline
Aminesaresyntheticintermediates but theyarerathertoxicchemicals. • Theirimportantreactions: Alkylation:
Acylation: (withacylchloridestogiveamides): Salt formation (withacids): Imineformation: (withaldehydesorketones):
Otherfunctionalgroupsconsisting of nitrogene Oximes R-CH=N-OH CH3-CH=N-OH C6H5-CH=N-OH acetaldehydeoximebenzaldehydeoxime (CH3)2C=N-OH acetoneoxime Nitrates (nitricacidesters) R-O-NO2 CH3-O-NO2methylnitrate C6H5-O-NO2phenylnitrate
Nitrites(nitrousacidesters) R-O-NO CH3-O-NO methylnitrite C6H5-O-NO phenylnitrite HydroxamicacidsR-CO-NH-OH Verytoxiccompounds These can be considered as N-hydroxiyamidesoracyl/aroylhydroxylamines C6H5-CO-NH-OH benzohydroxamicacidor N-hydroxybenzamide C6H5-CO-NH-OHbenzohydroxamicacidor N-hydroxybenzamide
Oxidation of amidesaredifficultcomparedtoamines but in case it is possible, hydroxamicacids can be obtained. Generallyaliphaticamidesaremuchmoreeasilyoxidized: HYDRAZINE DERIVATIVES Hydrazine is a verybasic, smallcompound H2N-NH2 Hydrazine can be alkylated, acylated as in thecase of aminestoormsomederivatives: NH2-NH-CH3 N-methylhydrazine
N-acylhydrazinesareknown as hydrazidesandtheseareimportantantimicrobialagents in medicinalchemistry: NH-NH-CO-CH3aceticacidhydrazide oracetichydrazide oracetohydrazide ISONIAZID (isonicotinicacidhydrazide) p-aminobenzoichydrazide
Ring hydrazide is alsoimportant: IMINES -C=N- is iminogroup N-nonsubstituted imine N-substituted imine Stabilityorder: N-aromaticsubstituted > N-aliphaticsubstituted > N-nonsubstitutedimines N-aromaticsubstitutediminesarecalled «SchiffBases»
CH3-CH=N-H 1-etanimine (CH3)2C=N-H 2-propanimine N-substitutediminesarenamed as N-alkylideneor N-arylidenesubstitutedamines: C6H5-N=CH-C6H5 N-benzylideneaniline CH3-N=CH-CH3 N-ethylidenemethylamine Imines can be easilyhydrolyzedbackto amine plusthecorrespondingcarbonylcompound (aldehydeorketone): R-N=CH-R + H2O -> R-NH2 + O=CH-R
HYDRAZONES Theseareproducts of hydrazineswithaldehydesandketones: R-CHO + H2N-NH2 -> R-CH=NH-NH2 (hydrazones) (R) (R) Thesecompoundshavegenerallyantimicrobialproperty. Nomenclature: CH3-CH=N-NH2acetalhydrazone (acetaldehydehydrazone) C6H5-CH=N-NH2benzalhydrazone (benzaldehydehydrazone)
NITRILE (CYANIDE) • Toxicmoiety • -CN cyanomoiety Theirnamescomefromthe name of thecorrespondingacids: CH3-CN acetonitrile (fromaceticacid) C6H5CN benzonitrile (frombenzoicacid) Alternatively, nitriles can be named as cyanides (namesaremethylcyanideorphenylcyanideforthecompoundsabove) Nitriles can be hydrolysedtocarboxylicacidsviaamides: RCN + H2O -> R-CONH2 R-CONH2 + H2O -> R-COOH
AZO COMPOUNDS R-N=N-R symmetrical R-N=N-R1asymmetrical CH3-N=N-CH3azomethane C3H7-N=N-C3H7azopropane CH3-CH2-N=N-CH3methylazoethane C6H5-N=N-CH3methylazobenzene (in assymetricones, thesmallercarbon is considered as a substituent)
AZOXY COMPOUNDS azoxybenzene N-NITROSAMINES N-nitrosoaniline C6H5-NH-NO NITRO COMPOUNDS R-NO2 CH3-NO2nitromethane C6H5-NO2nitrobenzene
NITROSO COMPOUNDS R-NO CH3-NO nitrosomethane C6H5-NO nitrosobenzene
NITRIC ACID ESTERS (NITRATES) R-O-NO2 CH3-O-NO2methylnitrate C6H5-O-NO2phenylnitrate NITROUS ACID ESTERS (NITRITES)R-O-NO CH3-O-NOmethylnitrite C6H5-O-NOphenylnitrite
NITRONES (Thisgroup can be considered as N-oxide of imines) N-ethyl-alpha-methyl-alpha-propylnitrone N,alpha-diphenylnitrone
UREA and DERIVATIVES HO-CO-OH carbonicacid NH2-CO-OH carbamicacid NH2-CO-NH2carbamide (urea) Urea is a monoamide of carbamicacidand a diamide of carbonicacid Differentnitrogenes can be namedeither N, N’ or N1, N3 andthese can be substitutedwithalkyl, arylorotherfunctonalgroups. NH2-CO-NH-CH3 (N-methylurea) Cl-NH-CO-NH-C2H5 (N-chloro-N’-ethylurea)
UREIDES (N-acylurea) NH2-CO-NH-CO-R NH2-CO-NH-CO-CH3 (N-acetylurea) Cyclicureide is alsoimportant in pharmaceuticalchemistry: Malonylurea (BARBITURIC ACID)
CARBAMATES (URETANE) NH2-CO-OR Carbamicacidestersarecalled as carbamatesoruretanes NH2-CO-OC2H5 is ethyluretaneorethylcarbamate
THIOUREA SEMICARBAZIDE (N-aminourea) NH2-CO-NH-NH2 1 2 3 4 SEMICARBAZONE (N-aminourea) NH2-CO-NH-NH=CH(R)-R NH2-CO-NH-NH=CH-CH3 (acetaldehydesemicarbazone)
THIOSEMICARBAZIDES (N-aminourea) NH2-CS-NH-NH2 1 2 3 4 THIOSEMICARBAZONES (N-aminourea) NH2-CS-NH-NH=CH(R)-R NH2-CO-NH-NH=CH-C6H5 (benzaldehydethiosemicarbazone)
IMPORTANT SULPHUR CONTAINING FUNCTIONAL GROUPS THIOALCOHOLS R-SH CH3-SH methanethiol THIOPHENOLS C6H5-SH THIOETHERS (SULFIDES) R-S-R CH3-S-CH3 (dimethylsulfide) C2H5-S-C6H5 (ethylphenylsulfide)
SULFOXIDES R-SO-R CH3-SO-CH3dimethylsulfoxide SULFONES R-SO2-R CH3-SO2-CH3dimethylsulfone
SULFONIC ACIDS R-SO3H CH3-SO3H methanesulfonicacid C6H5-SO3H benzenesulfonicacid
SULFONAMIDES R-SO2-NH2 Arylsulfonamidesareimportantantimicrobialagents. p-aminobenzenesulfonamide
SULFONYL CHLORIDE R-SO2-Cl Sulfonylchloridesareimportant SIM forproducingsulfonamides C2H5-SO2-Cl ethanesulfonylchloride C6H5-SO2-Cl benzenesulfonylchloride SULFONYLUREA R-SO2-NH-CO-NH2 Sulfonylureasareimportantantihyperglycemiccompounds: 1-butyl-3-p-(tolylsulfonyl)urea
THIONES Thionesare ring ketoneswith S (withthereplacement of O in ketones): Thiocyclohexanone (cyclohexanethione)
POLIFUNCTIONAL GROUPS Polyfunctionalgroupsmaycontainmorethanonefunctionalgroup in a molecule. One of them is selected as a main functionalgroupandthe rest is considered as substitents. Theorder of theirpriority is as follows: carboxylicacid > carboxylicacidderivatives > aldehydesandketones > alcohol > amine > doublebond Forexample: H2N-CH2-CH2-CH2-CH2-OH 4-amino-1-butanol (not 1-amino-4-butanol)
Unsaturatedacids CH2=CH-COOH 2-propenoic acid (acrylicacid) CH3-CH=CH-COOH 2-butenoic acid(crotonicacid) b. Unsaturatedaldehydes CH2=CH-COOH acrylicacid CH2=CH-CHO acrolein CH3-CH=CH-COOH crotonicacid CH3-CH=CH-COOH crotonaldehyde
c. Hydroxyacids CH3-CH(OH)-COOH lacticacid d. Amino acids NH2-CH2-COOH glycine CH3-CH(NH2)-COOH alpha-alanine H2N-CH2-CH2-COOH beta-alanine
e. Hydroxyaldehydes f. Polyoles (dioles, trioles) CH2OH CH2OH Ethylenglycol (GLYCOL) or 1,2-ethanediol CH2OH CHOH CH2OH Glycerol (GLYCERIN) or 1,2,3-propanetriol glucose
g. Diacids COOH COOH oxalicacid COOH CH2 COOH malonicacid COOH CH2 CH2 COOH succinicacid
IMPORTANT REACTIONS IN MEDICINAL CHEMISTRY • Alkylation (amines) • Acylation (amines) • Halogenation (aromaticsubstitution) • Esterification (carboxylicacid, alcohol) • Sulphonation (aromaticsubstitution) • Nitration(aromaticsubstitution) • Schiffbaseformation (imines, amines, aldehydes, ketones) • Oxidation (Aldehyde, ketone, alcohol, amine) • Reduction (aldehyde, ketone, nitro, azo,doublebond) • Hydrolysis (ester, amide, imine) • Decarboxylation (carboxylicacid) • Dealkylation (prandsec amine, ether, thioether)
1st groupsubstituents Theseareorthoand para directinggroupsie OH, NH2, X and CH3 Reactionsareeasiercomparedto 2nd groupsubstituents: 2,4,6-trichlorotoluene 2st groupsubstituents ThesearemetadirectinggroupsieCOOH, and NO2 groups Reactionsaremoredifficultcomparedto1st groupsubstituents: 3,5-dichloronitrobenzene