The Atom
The Atom. Essential Question?. Compare and contrast the characteristics of an atom and function of these characteristics in an atom?. Structure of an Atom. Proton p +. Positive Charge (p + ) Located in the nucleus Mass of a proton = 1.673 x 10 -24 g Relative Mass 1 amu

The Atom
E N D
Presentation Transcript
Essential Question? Compare and contrast the characteristics of an atom and function of these characteristics in an atom?
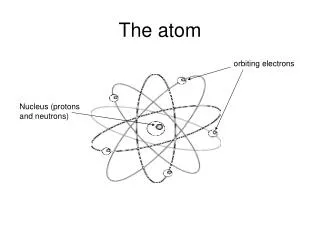
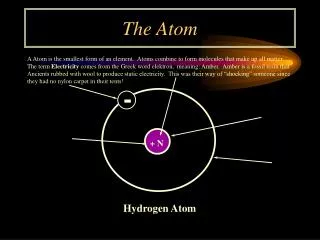
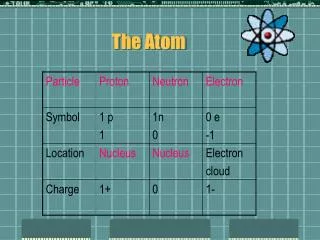
Protonp+ • Positive Charge (p+) • Located in the nucleus • Mass of a proton = 1.673 x 10-24g • Relative Mass 1 amu • Function: Gives an atom its identity – If the # of protons changes so does the type of atom Ex: Hydrogen has 1 proton Helium has 2 protons Lithium has 3 protons • The Atomic # always equals the # of protons Ex: Hydrogen has an Atomic # = 1 and has 1 proton Proton
Neutronno Neutron • No charge • Located in the nucleus • Mass of a neutron 1.675 x 10-24g • Relative Mass = 1 amu • Function: Adds mass to nucleus • Mass # - Atomic # = # of no Ex: 1-1=0 hydrogen has 0 no
Electrone- Electron • Negative charge • Has very small mass (9.109 x 10-28g) and is not included in the atomic mass of the atom • Relative Mass = 1/1840 • Located outside the nucleus - electron cloud - orbitals - shells - energy levels Ex: Hydrogen has 1e- • Function: Is responsible for an atoms behavior
Atomic # = # protons = # electrons • Which element has 32 protons? Symbol__ Name __ • Which element has 14 protons? Symbol__ Name __ • The mass number is the number of protons PLUS the number of neutrons in an atom. • This is because the mass of the atom is mainly inside the nucleus where the protons and neutrons are found. • The mass number – protons = number of neutrons on the Periodic Table
This will help you determine which type of atom you have • If the p+≠ e- then you have an ION • If you add the p+ + no & the mass # ≠ the mass on the PT, you have an ISOTOPE • If the mass # = the mass # on the PT, and the p+ = e- then you have an Element
Element • Cannot be broken down into simpler substances by physical or chemical means • Each has a symbol located on the Periodic Table Ex: Ag – Silver, Pb – Lead • Each has an atomic # and atomic mass Ex: Ag – Atomic # = 47 Atomic mass = 107.9 amu Pb – Atomic # = 82 Atomic mass = 207.2 amu • Atomic # = # of p+ = # of e- • If the # of p+ = the # of e- the atom is electrically neutral • Atomic mass = # of p+ + # of no (this number must match the rounded atomic mass (or Mass #) on the periodic table)
Ion • An ion is an atom with a charge. The charge is either positive or negative • The charge depends on if the electrons are donated (lost) or accepted (gained) Ex: If an atom donates e- then the charge becomes positive (+) Na+ Ex: If an atom accepts e- then the charge becomes negative (-) Cl- • To identify an ion the # of protons will NOT equal the # of electrons Ex: Cl normally has 17e- A Cl- ion has 18e- (Cl has accepted 1e-)
Ion - Fill in the table below Are the number of protons and electrons the same or different? DIFFERENT
Isotopes • Are atoms with the same number of protons but has a different number of neutrons than the normal number of neutrons in an atom • To calculate the # of no round the atomic mass to the nearest whole number then subtract the atomic number. • When the atomic mass is rounded it is called the Mass number Mass number–Atomic number =# of no Ex: (Pb) 207 – 82 = 125 neutrons • If the atom is an isotope then there will be more neutrons and the mass will be more Ex: (Pb) 208 –82 = 126 neutrons • Isotopes are neutron rich and unstable
Isotopic Notation Ex: Silicon Mass # 28 Si 14 Atomic # Isotopic Name Silicon-28 Mass #
Let’s Compare the Following How are the notations alike? How are the above notations different? Same protons and electrons Different Mass # & neutrons
Review Isotopes are atoms of the same element with the same number of _______, but with different numbers of _______. Answer questions 1-5 in Part I Answer questions 6-10 in Part II
Summarize • Fill in Part V chart
Summarize • Answer questions part VI
Part VII Potassium • K – 39 has 20 neutrons • K – 40 has 21 neutrons • K – 41 has 22 neutrons
Atomic Mass Determination • Chemist have developed a method of measuring mass of an atom called: Atomic Mass Determination Unit is abbreviated “amu” • They did this because the masses of subatomic particles are so small that even scientific notation makes it hard to calculate the masses of elements. • The atomic mass that you see on the PT is the average mass for the isotopes of that element. • The atomic mass you see on the PT is also the isotope that is most abundant in nature!!! • Answer Question 1 & 2
How is Atomic Mass Calculated? • The mass on the Periodic Table is the average mass of the isotopes of an atom • To determine the atomic mass of an atom use this formula (Mass (amu) x Percent abundance)= Isotope A (Mass (amu) x Percent abundance)= Isotope B Add A + B / 100 = atomic mass
Example Problem #1 • Calculate the atomic mass of the unknown element. Then identify the element using the Periodic Table?
(184.953 amu x 37.40) = 6917.35 amu (186.956 amu x 62.60) = 11703.45 amu 6917.35 amu trace/least abundant +11703.45 amu most abundant 18620.8 / 100 = 186.2 amu Rhenium = Re
Practice Problem #2 • Calculate the atomic mass of the unknown element. Then identify the element? Which Isotope is trace and which is more abundant?
(112.904 amu x 4.30 ) = 485 amu (114.904 amu x 95.70) = 10996.3128 amu 485 amu trace/least abundant +10996.3128 amu most abundant 11481.3128 / 100 = 114.81 amu Indium = In
Practice Problems (Book) • Pp. 104 15-17 #15Boron has 2 naturally occurring isotopes: boron-10(abundance = 19.8%, mass = 10.013 amu), boron-11 (abundance = 80.2 %, mass = 11.009 amu) Calculate the atomic mass of boron. #16 Helium has 2 naturally occurring isotopes, helium-3 and helium-4. The atomic mass of helium is 4.003 amu. Which isotope is more abundant in nature? Explain #17 Calculate the atomic mass of magnesium. The three magnesium isotopes have atomic masses and relative abundances of 23.995 amu (78.99%), 24.986 amu (10.00%), 25.982 amu (11.01%).
Practice Problem #1 Answer = 10.81 amu 5 5
Practice Problem #2 Answer = 107.9 amu 47 47
Practice Problem #3 Answer = 24.31 amu 12 12 12
Practice Problem #4 Helium-4 is most abundant b/c its mass is closer to the average atomic mass on the PT Helium-3 mass is not as close as Helium-4, so Helium-3 is less abundant
Practice Problem #5 Answer = 20.19 amu 10 10
Practice Problem #6 Answer = 178.55 amu 72 72 72 72 72
Practice Problem #7 Answer = 28.09 amu 14 14 14
Practice Problem #8 Answer = 107.87 amu 47 47
Practice Problem #9 Answer = Neon-20 Why? Its mass is closest to the average mass of neon on the PT.
Practice Problem #10 Answer = 83.80 amu 36 36 36 36 36 36