cARBOHYDRATE
cARBOHYDRATE. Kuiah-3. Elements: C, H, and O. Definisi . Karbohidrat (' hidrat dari karbon ', hidrat arang ) atau sakarida (dari bahasa Yunani σάκχαρον , sákcharon, berarti " gula ") adalah segolongan besar senyawa organik yang paling melimpah di bumi.


cARBOHYDRATE
E N D
Presentation Transcript
cARBOHYDRATE Kuiah-3 Elements: C, H, and O
Definisi • Karbohidrat ('hidrat dari karbon', hidrat arang) atau sakarida (dari bahasa Yunaniσάκχαρον, sákcharon, berarti "gula") adalah segolongan besar senyawa organik yang paling melimpah di bumi. • Karbohidrat memiliki berbagai fungsi dalam tubuh makhluk hidup, terutama sebagai bahan bakar (misalnya glukosa), cadangan makanan (misalnya pati pada tumbuhan dan glikogen pada hewan), dan materi pembangun (misalnya selulosa pada tumbuhan, kitin pada hewan dan jamur).Pada proses fotosintesis, tetumbuhan hijau mengubah karbon dioksida menjadi karbohidrat.
Definisi • Secara biokimia, karbohidrat adalah polihidroksil-aldehida atau polihidroksil-keton, atau senyawa yang menghasilkan senyawa-senyawa ini bila dihidrolisis. • Karbohidrat mengandung gugus fungsikarbonil (C=O) (sebagai aldehida (RCHO) atau keton , RCOR’) dan banyak gugus hidroksil (-OH). Pada awalnya, istilah karbohidrat digunakan untuk golongan senyawa yang mempunyai rumus (CH2O)n, yaitu senyawa-senyawa yang n atom karbonnya tampak terhidrasi oleh n molekul air.Namun demikian, terdapat pula karbohidrat yang tidak memiliki rumus demikian dan ada pula yang mengandung nitrogen, fosforus, atau sulfur
Gugus dalam KH Gugus Keton (RCOR’) Gugus karbonil (C=O) Gugus Aldehid (R=CHO)
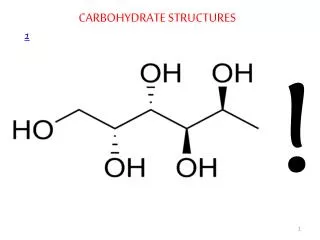
Structure • Carbohydrates are organic compounds that usually contain carbon hydrogen and oxygen in the ratios: 1 Carbon: 2 Hydrogens: 1 Oxygen, (CH2O)n
Class of Carbohydrate • There are four classes of carbohydrates that are of general interest: • Monosaccharides, (3 – 9 atom karbon) • Disaccharides, (2 molekul monosakarida) • Oligosaccharides, (beberapa monosakarida) • Polysaccarides (karbohidrat kompleks, banyak sekali monosakarida)
Gabungan monosakarida • Gabungan senyawa2 monosakarida membentuk senyawa karbohidrat yang lebih besar. Ikatan penghubung antara monosakarida disebut ikatan glukosida • Disakarida 1 ikatan glukosida • Trisakarida 2 ikatan glukosida • Dst
Klasifikasi karbohidrat • Ada dua jenis monosakarida: • Aldosa mengandung gugus aldehid • Ketosa menganung gugus keton • Monosakarida juga dapat dikelompokkan menurut jumlah atom karbon: • Triosa (3 atom karbon) • Tetrosa (4 atom karbon) • Pentosa (5 atom karbon) • Hexosa (6 atom karbon)
Bentuk molekul karbohidrat ketotetrose aldotetrose ketopentose aldopentose
Monosaccharide • Monosaccharides (simple sugars) have a carbon skeleton of 3 or more carbons depending on the monosaccharide. The most familiar monosaccharide is Glucose (C6 H12 O6). • A ball and stick model of glucose is shown here in its ring form, which is the form it takes in water. As a solid, glucose has a straight chain form which is not shown.
D-glucose is an aldohexose with the formula (C·H2O)6. The red atoms highlight the aldehyde group, and the blue atoms highlight the asymmetric center furthest from the aldehyde; because this -OH is on the right of the Fischer projection, this is a D sugar. Glucose can exist in both a straight-chain and ring form.
Galactose • Galactose is another monosaccharide with six carbons. Later we will meet fructose or fruit sugar which also has six cabons. • Galacotse is a component of a disaccharide called lactose
Fructose Glucose Galactose Three common monosaccharide hexoses (simple structure)
Two common monosaccharidespentoses (ring structure) Two common pentoses found in living organisms are ribose and deoxyribose. Ribose and deoxyribose differ in that ribose has a hydroxyl group attached to its 2nd carbon, while deoxyribose has two hydrogens. Deoxyribose and ribose are the main structure of genetic materials –called gene
Disaccharides • Two sugars bonded together for the next simplest sugar, disaccaharides (di–: two; –saccaharides: sugars). • Two common disaccharides are lactose, milk sugar, and sucrose, table sugar. • Lactose is found in mammals milk, made in the mammary glands from glucose and galactose. • Sucrose is found in fruits vegetables, and honey, and is made from glucose and fructose.
Disaccharides. Disaccharides consist of two monosaccharides linked together by a dehydration synthesis. • Sucrose is common disaccharide which functions as a transport sugar in plants. The production of sucrose by means of a dehydration synthesis is shown here. Each sucrose molecule is made by chemically combining a glucose and a fructose molecule. • A hydrogen is removed from the glucose and a hydroxyl(OH) from the fructose leaving an oxygen to link the two molecules together. • Lactose, another disaccharide, is commonly called milk sugar. • This diagram shows the synthesis of sucrose from glucose and fructose via a dehydration synthesis.
Lactose is contructed from glucose and galactose Sucrose is contructed from flucose and fructose
Oligosaccharides • Oligo means a few and oligosaccharides have a few simple sugars linked together but not • Oligosacharides are common on cell membranes and surfaces where they often serve as cell markers.
Polyscharides • The largest carbohydrates are polysaccharides (poly–: many; –saccaharides: sugars). • Polysaccharides are long chains of sugars, bonded together. Two common polysaccharides are starch and cellulose. • Both polysaccharides are made from long chains of glucose, but they differ in how those glucose molecules are bonded together. • In starch, glucose are linked by an a (1-4) bond, while in cellulose, glucose are linked by a b (1-4) bond. • In both, the 1st carbon of one glucose is bonded to the 4th carbon of the next glucose molecule. But how those carbons bond differ by where they hold onto each other. • Not all polysaccharides are made merely of chains of sugars. Some, like glycogen, have many branches of sugars running off of a main branch.
Three common polysaccharides (ring structure) Starch / Amylose Cellulose Glycogen
Function • Polysaccharides provide mid-term energy storage and structural components to organisms. • Starch is used to store energy in plant cells. In plant cells, you can seen starch granules in some organelles. • Glycogen forms similar midterm storage in animals. In mammals, glycogen takes up 5% of the liver by weight, and 0.5% of muscles by weight. • Cellulose is a sturdy molecule that’s insoluble in water, forming the main structural fibers for plant cells' walls.
Carbohydrates are sugars and starches. The most basic structure consist of 3-6 carbons, but we are going to concentrate upon sugars that form a either a pentagon ring (5-carbon sugars) or a hexagon ring (6-carbon sugars). These sugars are named pentoses and hexoses respectively. The sufix –oses refers to sugar, and prefix refers to the number of carbons. One corner of the ring has an oxygen, so that one carbon group lies outside of the ring. Attached to each carbon is a hydroxyl group, and a hydrogen. If you said that carbohydrates had a primary structure, akin to proteins, it would be the order of the sugars, the pentoses and the hexoses. The simplest sugars are monosaccharides (mono–: one; –saccaharides: sugar). Among the hexoses, sugars having six carbons, there are glucose, galactose, and fructose. Both glucose and galactose have very similar structures, and only differ in the arrangement of on hydroxyl group on the 4th carbon. Fructose looks more like glucose than galactose, but it differs from glucose by having a hydroxyl group on the 1st carbon, with its 2nd carbon having the double bond with oxygen.