Electrolytic Properties of Aqueous Solutions
Explore the concept of dissociation, solution conductivity, and Arrhenius’s theory of electrolytic dissociation. Learn about different types of electrolytes and how they affect solution conductivity in this interactive guide.

Electrolytic Properties of Aqueous Solutions
E N D
Presentation Transcript
Guiding Questions What is meant by dissociation? What is the molecular arrangement of water molecules?
A. Definitions • Solution - homogeneous mixture Solute - substance being dissolved Solvent - present in greater amount
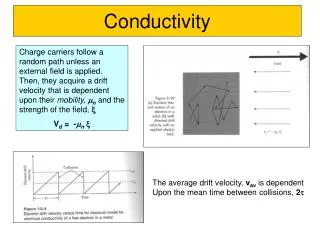
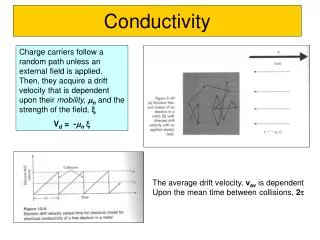
Arrhenius’s Theory ofElectrolytic Dissociation • Why do some solutions conduct electricity? • An early hypothesis was that electricity produced ions in solution, and those ions allowed the electricity to flow. • Arrhenius’s theory: • Certain substances dissociate into cations and anions when dissolved in water. • The ions already present in solution allow electricity to flow.
Dissociation • http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/molvie1.swf
Nonpolar vs. Polar • When nonpolar substances dissolve in one another, they simply mix. • When polar substances dissolve, electrical reactions pull apart the molecules.
K+(aq) + MnO4-(aq) IONIC COMPOUNDSCompounds in Aqueous Solution Many reactions involve ionic compounds, especially reactions in water — aqueous solutions. KMnO4 in water
ELECTROLYTES • Are substances that form positive(+) and negative (-) ions in water • Conduct an electric current
Types of Electrolytes • A strong electrolyte dissociates completely. • A strong electrolyte is present in solution almost exclusively as ions. • Strong electrolyte solutions are good conductors. • A weak electrolyte dissociates partially. • Weak electrolyte solutions are poor conductors. • Different weak electrolytes dissociate to different extents. • A nonelectrolyte does not dissociate. • A nonelectrolyte is present in solution almost exclusively as molecules. • Nonelectrolyte solutions do not conduct electricity.
Electrolytic Properties of Aqueous Solutions • Electrolytesdissociate to produce ions. The more the electrolyte dissociates, the more ions it produces.
Is it a strong electrolyte, a weak electrolyte, or a nonelectrolyte? • Strong electrolytes include: • Strong acids (HCl, HBr, HI, HNO3, H2SO4, HClO4) • Strong bases (IA and IIA hydroxides) • Most water-soluble ionic compounds • Weak electrolytes include: • Weak acids and weak bases • A few ionic compounds • Nonelectrolytes include: • Most molecular compounds • Most organic compounds (most of them are molecular) How do we tell whether an acid (or base) is weak?
- + - - + + acetic acid salt sugar B. Solvation Non- Electrolyte Weak Electrolyte Strong Electrolyte solute exists as ions and molecules solute exists as ions only solute exists as molecules only DISSOCIATION IONIZATION View animation online.
Electrolytes in the Body • Carry messages to and from the brain as electrical signals • Maintain cellular function with the correct concentrations electrolytes