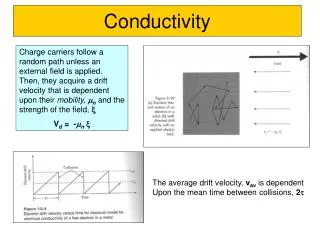
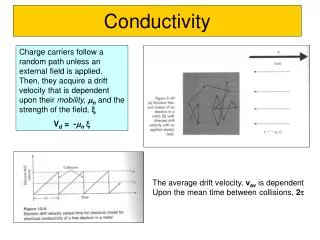
Conductivity
Conductivity. A Conductor is a substance that allows electricity to pass through it. An Insulator is a substance that will not allow electricity to pass through it. An Electric current is a flow of Ions through a Liquid. An Electric current is a flow of electrons through a Solid.

Conductivity
E N D
Presentation Transcript
Conductivity • A Conductor is a substance that allows electricity to pass through it. • An Insulator is a substance that will not allow electricity to pass through it. An Electric current is a flow of Ions through a Liquid An Electric current is a flow of electrons through a Solid
Conductivity and Bonding • There are three main types of Bonding we shall consider here: ` 1.Metallic 2.Ionic3.Covalent. • Metallic Bonding:This can be regarded as metallic atoms which are so close to each other that their outer electrons can move freely between atoms hence causing an electric current. • All METALS Conduct electricity
Conductivity and Ionic Bonding • Ionic Bonding: Occurs between Metal and Non-Metal atoms. An Ionic compound conducts Electricity when Molten or in Solution since the Lattice is broken down and theIons are free to move When in solid form an Ionic Compound arranges itself in a special structure called a Lattice + = Na+ion - =Cl-ion Ionic compounds do not Conduct when Solid since the Ions are not free to move
Conductivity and Covalent Bonding • Covalent Bonding: Occurs between non-metal Atoms • Covalent substances contain no Ions and therefore do not conduct electricity when solid , molten or in solution. • Carbon(in the form of Graphite) is an exception to the above rule. It has a special type of Covalent Bonding called a Covalent Network.
Electrolysis • Electrolysis is the passing of an Electric current through a substance so that it breaks up at each Electrode Electrolysis of Copper Chloride 6 volt dc.power supply.It has to be a dc.current so that the current flows only in one direction -ve.electrode +ve.Electrode Electrodes are made of graphite which are good conductors but don’t easily take part in the reaction
Electrolysis explained The diagram below is a result of the fact that opposite charges attract.On reaching each electrode they lose or gain electrons and become Elements in their natural form +ve electrode -ve electrode Cu2+ Cl- Cu2+ Cl- Cu2+ Bubbles of Chlorine gas are formed here Copper metal is formed here Cl-
At the -ve electrodeCu2+ ions receive two electrons and change to Cu metal.Since they gain electrons we say that they have been REDUCED.ie Reduction takes place at the –ve electrode. At the +ve electrode Cl- ions lose an electron and change to Cl atoms.We say that they have been OXIDISED. Now since chlorine is Diatomicit waits for another Ion to do the same so that it can be given off as Cl2 molecules(gas) .Oxidation takes place at the +ve electrode Ion Electron Half-Equations Cu2+ + 2e Cu 2Cl- Cl2 + 2e Reduction Oxidation To get the Redox or Ionic Equation we simply “ADD” the Oxidation and Reduction Equations together Cu2+ + 2e Cu Reduction 2Cl- Cl2 + 2e Oxidation Cu2+ + 2Cl- Cu +Cl2 IONIC
Solubility • The rule for SOLUBILITY is that: Like dissolves Like This means that the Substance you are trying to dissolve must have the same type of Bonding as the liquid you are trying to Dissolve it in. eg Covalent substances dissolve in Covalent liquidsIonic substances dissolve in Ionic liquidsNote: most Ionic substances also dissolve in water
Melting Points and Boiling Points • Ionic substances when Solid, exist in very stable structures called Lattices which form strong Ionic bonds between positive and negative Ions. Therefore to break these up we require a lot of Energy and hence the Melting Pts and Boiling Pts. are high. • Covalent substances have weaker bonds than Ionic substances and have only very weak bonds between molecules(called Van der Waals forces).As a result not as much energy is required to break up these substances and hence Melting Pts and Boiling Pts.are low. An exception to this rule is Covalent Networks,such as, Graphite and Silicon Dioxide who form large stable structures which are difficult to break up and therefore have High Mpts.&Bpts
Colour of Ions • Whilst most Ions are colourless, some are brightly coloured: • eg.Cu2+ Ni2+ Dichromate Permanganate Chromate When these Ions form a compound with another colourless Ion then the compound will be the colour of the coloured Ion.When two coloured Ions join, the compound is a mixture of the two of the two colours eg.Copper Chromate is Green