lipids
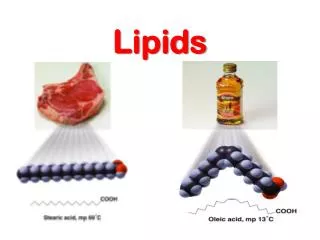
lipids. Lipids are water insoluble, oily or greasy organic compounds soluble in non-polar organic solvents. Chemically, lipids are defined as the esters of alcohol and fatty acids. Lipids are composed of 3 fatty acids joined to an alcohol .


lipids
E N D
Presentation Transcript
lipids • Lipids are water insoluble, oily or greasy organic compounds soluble in non-polar organic solvents. • Chemically, lipids are defined as the esters of alcohol and fatty acids.
Lipids are composed of 3 fatty acids joined to an alcohol. • Fatty acids and alcohol are the building block components of lipids. • The lipids are the important constituents of diet due to their higher energy value. • One gram of lipid yields 9.3 kilocalones of heat, while the same amount of carbohydrate or protein yields 4.5 kilocalories only.
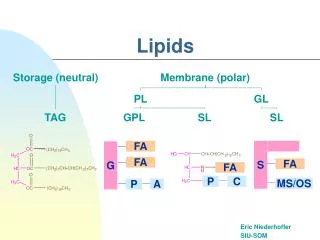
The lipids include a heterogenous group of compounds related to fatty acids. • The common lipids are fats. oils, waxes, phosph olipids, glycolipids, cerebros ides, sulfolipids, aminolipids, steroids, terpenes, carotenoids, some hormones and some vitamins.
Lipids have three important properties. 1. Lipids are insoluble in water but soluble in non-polar organic solvents, such as acetone, alcohol, chloroform, benzene and ether. • 2. They contain a large proportion of carbon and hydrogen bonds and release large amount of energy On breakdown. • 3. On alkaline hydrolysis lipids yield alcohol and fattyacids.
Lipids are esters* of glycerol and fatty acids. They are formed by the combination of alcohol and fatty acids. • Usually a lipid is made up of a glycerol and three fatty acids. Such a lipid is called a triglyceride or a neutral fat.
Ester is a compound formed by the combination of an acid with a glycerol with the removal of water.
Glycerol, a component of lipid. It is an alcohol. It is a trihydric alcohol. It contains three alcoholic (OH) groups. Of these three, two are primary alcoholic groups (CH2OH) and the third one is secondary (CHOH).
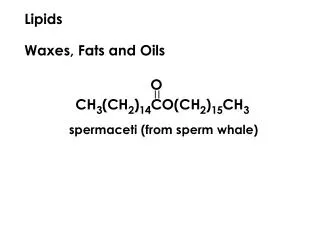
Fatty acid: A long-chain carboxylic acid; those in animal fats and vegetable oils often have 12–22 carbon atoms. • Lipid: A naturally occurring molecule from a plant or animal soluble in nonpolar organic solvents. • Waxesare carboxylic acid esters, RCOOR’,with long, straight hydrocarbon chains in both R groups; they are secreted by sebaceous glands in the skin of animals and perform mostly external protective functions.
Triacylglycerols are carboxylic acid triesters of glycerol, a three-carbon trialcohol. They make up the fats stored in our bodies and most dietary fats and oils. They are a major source of biochemical energy.
Fatty Acids and Their Esters • The naturally occurring fats and oils are triesters formed between glycerol and fatty acids. • Saturated fatty acid: A long-chain carboxylic acid containing only carbon–carbon single bonds. • Unsaturated fatty acid: A long-chain carboxylic acid containing one or more carbon–carbon double bonds. • If double bonds are present in naturally occurring fats and oils, the double bonds are usually cis rather than trans.
Trans Fatty Acids The Truly Awful!
The Good!
The Bad!
The Truly Awful!
Strcture of lipids The figure is found at http://courses.cm.utexas.edu/archive/Spring2002/CH339K/Robertus/overheads-2/ch11_lipid-struct.jpg(Jan 2007)
Glycerides The major form of lipid in food and in the body
The figure was adopted from http://en.wikipedia.org/wiki/Fatty_acid (April 2007)
Fatty Acids • Fatty acids are aliphatic straight chain hydrocarbon compounds with a terminal carboxyl group. They are the building blocks of lipids.
The carbon atoms are numbered from the carbon of the COOH group. • In most of the unsaturated fatty acids there is a single double bond lying between carbon atoms 9 and 10. • This is designated as . • The symbol with the superscript number • 9 indicates the position of the double bond.
The symbol 18:0 denotes a C,8 fatty acid with no double bonds. The number 18:2 signifies that there are 2 double bonds. • Similarly, the symbol 18:2; 9, 12 is used to denote an 18 carbon acid with two double bonds in the 9 and 12 positions. • When two or more double bonds are present in a fatty acid, the double bonds are never conjugated But the double bonds are separated by a methylene group.
1- Simple lipids • Neutral fat (Triacylglycerol): • Esters of 3 fatty acids with glycerol • Glycerol is an alcohol containing 3 OH groups • Types: • Simple: contain similar fatty acids e.g., tripalmitin • Mixed; contain different fatty acids e.g., palmito-oleio-stearin • Either solid, called fats, or liquid, called oils. • Waxes: • Esters of fatty acids with alcohols other than glycerol. • Important for manufacturing of ointments & cosmetics • Not digested
Simple Lipids (Triglycerides) • A simple lipid is formed when three molecules of fatty acids combine with one molecule of glycerol. • In this process 3 molecules of water are released.
Monoglycerides monopalmitin • Diglycerides dipalmitin
Polyunsaturated fatty acids have more than one C=C double bond. Linoleic and linolenic acids are essential in the human diet because the body does not synthesize them and they are needed for the synthesis of other lipids.
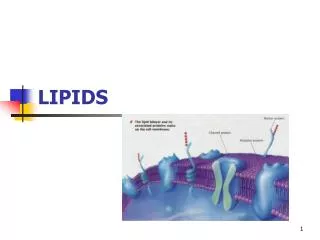
Compound lipids • contain some chemical groups in addition to fatty acids and glycerol. • When a lipid contains a phosphate group, it is called a phospholipid and when a lipid contains a carbohydrate, it is called a glycolipid.
Lecithin is a phospholipid. In lecithin, two hydroxyl groups of glycerol have been esterified by fatty acids, while the third hydroxyl group has been replaced by phosphoric acid which in turn has formed an ester with choline.
Phospholipid Lecithin, a common food additive, is a phospholipid. Embedded in cell membranes.
Derived lipids • Steroids: • The steroids have a 1,2_cycIopentanoperhydro phenanthrene nucleus. It has four rings named as A, B, C and D. The rings A, B and C are hexagons called cyclohexane rings. • The ring D is a pentagon called cyclopentane.
Steroids • Steroids are solid alcohols. • They have a cyclo pentano per hydro phenanthrene nucleus. • They are made up of four rings named as A,B,C and D. • The rings A, B and C are cyclohexanes. The ring D is a cyclopentane. The numbering starts from ring A to D.
it has two methyl groups (CH3 ) at carbon atoms 10 and 13. Usually there is a side chain at position 17. Cholesterol is a steroid.
Terpenes • derivatives of isoprene(= 2-methylbuta-1,3-diene) • found in oils of plants and flowers • characteristic odour (geraniol, menthol,...) • steroids are derived from triterpenes
Terpenes • Terpenes are hydrocarbons. They have less than 40 carbon atoms. • Terpenes are constructed out of isoprene units. • Each isoprene unit has 5 carbon atoms and 8 hydrogen atoms (C5H8). Each isoprene unit has two ends, namely a head and a tail. The head is the branched end and the tail is the unbranched
Terpenes – classification: • monoterpenes (C10) 2 x isoprene • sesquiterpenes (C15) 3 x isoprene • diterpenes (C20) 4 x isoprene • triterpenes (C30) 6 x isoprene • tetraterpenes (C40) 8 x isoprene • formed by bonding „head to tail“ or „tail to tail“ • different degree of unsaturation • variety of functional groups
When the terpene contains two isoprene units, it is called a monoterpene, When it contains 3 units, it is called a sesquiterpene. • When it contains 4 units the terpene is called • a diterpene. • A triterpene contains 6 units. • When there are 8 units the terpene is called a tetraterpene. • A polyterpene contains more than 8 isoprene units.
Examples of terpenes menthol (C10) phytol (C 20) squalene (C 30) -carotene (C40) The figures are adopted from http://en.wikipedia.org (April 2007)
The commercial name for glycerol is glycerine, • It soluble in water and insoluble in organic solvents. • Glycerol combines with three similar fatty acids to form a simple lipid called triglyceride with the release of 3 molecules of water. Glycerol has two important properties. They are 1. Formation of esters 2. Dehydration
1. Formation of Esters • Glycerol reacts with acids, both organic and inorganic acids, to form esters like monoesters, diesters and triesters, • Triesters of glycerol with higher fatty acids constitute lipids • Monoglycerides act as good detergents and emulsifying agents. This property helps in the manufacture of detergents. In animals, emulsifying property helps the digestion of fats. • 2. Dehydration • When glycerol is heated in the presence of a dehydrating agent like H2S04, phosphorus pentoxide or potassium hydrogen sulphate (KHSO4), it produces an unsaturated aldehyde called acrylic aldehyde or acrolein.
The common higher fatty acids are insoluble in • water. • But they can be dispersed into micelles in dilute NaOH Or KOH. • Micelle is an aggregation of fatty acid molecules • In water into a globular structure in which their non-polar • tails are in the interior and the polar heads are on the exterior exposed to water. • NaOH or KOH converts fatty acids into soaps. Soaps are the salts of fatty acids.
Again, based on their requirement in the diet, fatty • acids are classified into two types namely • 1. Essential fatty acids • 2. non- essential fatty acids • Thus fatty acids are classified into 6 types. They are • 1.unsaturated fatty acids • 2. saturated fatty acids • 3. hydroxy or oxygenated fatty acids • 4. Cyclic fatty acids • 5. essential fatty acids • 6. Nonessential fatty acids