Solubility Rules and Precipitation Reactions in Chemistry
Learn about solubility rules, metathesis reactions, and oxidation states in chemistry. Understand how to predict precipitate formation and identify spectator ions.

Solubility Rules and Precipitation Reactions in Chemistry
E N D
Presentation Transcript
Solubility Rules #2 Rule #1 Rule [Exceptions]
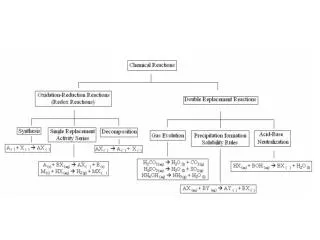
Metathesis (Double Displacement) AX + BYAY + BX Acid-Base neutralization A=H+ X=anion of the acid BY=Base HCl + NaOHH2O + NaCl note: In the case of ammonia: AX + YAY + X HCl+NH3NH4+ +Cl-
Precipitation Reactions (metathesis) AX + BYAY(s) + BX AX=Soluble Salt BY=Soluble Salt AY=Insoluble Solid (precipitate,ppt) BX=Soluble Salt AgNO3 + NaClAgCl(s) + NaNO3 Formula unit Ag++NO3- + Na+ + Cl-AgCl(s) + Na+ + NO3- Total Ionic Ag++ Cl-AgCl(s) Net Ionic Spectator Ions = nitrate ions and sodium ions
Fe(NO3)3 + 3NaOH Fe(OH)3(s) + 3NaNO3 Fe3+ + 3NO3- + 3Na+ + 3OH- Fe(OH)3(s) + 3NO3- + 3Na+ Fe3+ + 3OH- Fe(OH)3(s)
Will a precipitate form if we add solid Calcium Nitrate and solid Potassium Carbonate to water? Ca(NO3)2 Ca2+ + 2NO3- K2CO3 2K+ + CO32- Ca2+ NO3- K+ CO32- Ca(NO3)2 + K2CO3 CaCO3(s) + 2KNO3 Formula Unit Ca2+ + 2NO3- + 2K+ + CO32- CaCO3(s) + 2NO3- + 2K+ Total Ionic Ca2+ + CO32- CaCO3(s) Net Ionic Spectator ions: nitrate ions and potassium ions
Oxidation State (Oxidation Number) 2Na(s) 2Na+(aq) + 2e- oxidation 1/2 reaction 2H2O + 2e- 2OH-(aq) + H2(g) reduction 1/2 reaction 2Na(s)+2H2O2NaOH(aq)+H2(g) redox reaction oxidation-loss of electrons reduction-gain of electrons reducing agent-compound that is oxidized (Na(s)) oxidizing agent-compound that is reduced (H2O) We need an accounting system-oxidation state
Oxidation State Rules • The oxidation state of any atom in a free uncombined element is zero (Cl2, H2, O2, P4, Na(s) ... ) • The oxidation state of any monatomic ion is equal to the charge (Na+ ox. state = +1 ; Cl- ox. state = -1 ; Fe2+ ox. state = +2) • The sum of oxidation numbers of all atoms in a compound is zero • The sum of oxidation numbers of all atoms in an ion is equal to the charge of the ion (SO42- sum of ox. numbers = -2)
1 2 3 4 5 6 7 8
-2 +1 0 -2 2Na(s) + 2H2O2NaOH(aq) + H2(g) +1 +1 0 H2O 2*(+1) + X = 0 X= -2 for O in H2O NaOH (+1) + (+1) + X = 0 X=-2 for O in NaOH OH-- (+1) + X = -1 X=-2 for O in OH- Oxidation-increase in the oxidation state (number) Na(s) 0 +1 Reduction-decrease in the oxidation state (number) H +1 0