THE HEART
THE HEART. Normal Pathology Heart Failure: L, R Heart Disease Congenital : L R shunts, R L shunts, Obstructive Ischemic : Angina, Infarction, Chronic Ischemia, Sudden Death Hypertensive : Left sided, Right sided

THE HEART
E N D
Presentation Transcript
THE HEART • Normal • Pathology • Heart Failure: L, R • Heart Disease • Congenital: LR shunts, RL shunts, Obstructive • Ischemic: Angina, Infarction, Chronic Ischemia, Sudden Death • Hypertensive: Left sided, Right sided • Valvular: AS, MVP, Rheumatic, Infective, Non-Infective, Carcinoid, Artificial Valves • Cardiomyopathy: Dilated, Hypertrophic, Restrictive, Myocarditis, Other • Pericardium: Effusions, Pericarditis • Tumors: Primary, Effects of Other Primaries • Transplants
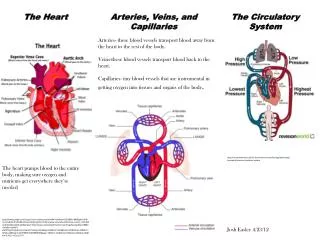
NORMAL Features • 6000 L/day • 250-300 grams • 40% of all deaths (2x cancer) • Wall thickness ~ pressure • (i.e., a wall is only as thick as it has to be) • LV=1.5 cm • RV= 0.5 cm • Atria =.2 cm • Systole/Diastole • Starling’s Law
Epicardial Coronary Arteries Chambers Increased left atrial cavity size Tortuosity Decreased left ventricular cavity size Increased cross-sectional luminal area Sigmoid-shaped ventricular septum Calcific deposits Atherosclerotic plaque Myocardium Valves Increased mass Increased subepicardial fat Aortic valve calcific deposits Brown atrophy Mitral valve annular calcific deposits Lipofuscin deposition Fibrous thickening of leaflets Basophilic degeneration (glyc.) Buckling of mitral leaflets toward the left atrium Amyloid deposits CARDIAC AGING
Aorta Dilated ascending aorta with rightward shift Elongated (tortuous) thoracic aorta Sinotubular junction calcific deposits Elastic fragmentation and collagen accumulation Atherosclerotic plaque CARDIAC AGING
BROWN ATROPHY, HEART LIPOFUCSIN
Pathologic Pump Possibilities • Primary myocardial failure (MYOPATHY) • Obstruction to flow (VALVE) • Regurgitant flow (VALVE) • Conduction disorders (CONDUCTION SYSTEM) • Failure to contain blood (WALL INTEGRITY)
Heart Failure • Definition--The heart is unable to pump blood at a rate that meets the requirements of the metabolizing tissues, or can only do so only with filling pressures that are higher than normal. • Onset may be insidious or acute.
Types • High output versus low output • Forward failure • Backward failure • Systolic failure • Diastolic failure
Important • Left heart failure and • Right heart failure
Physiological mechanism try to componsate heart failure • 1.The frank starlings mechanism • Increased filling volume dilate the ventricle • Increases the contractility • 2.Ventricular remodeling with or with out dilation • The structural molecular and cellular changes occur in ventricles due to overload is called ventricular remodelying.
3.Activation of neurohumoral system • The release of nroepinephrine increases the heart rate and increases the contractility • Activation of renin and angiotesnogenaldosterone mechanism • Release of atrialnatriuritic peptides
With all these the heart will be in a state of compensatory failure • Decompensate failure due to worsening abnormalities state is when heart is unable to bear • Myocardial structural changes, including augmented muscle mass (hypertrophy
Concentric hypertrophy, In pressure overload states the hypertrophy is characterized by increased diameter of individual muscle fibers. The thickness increases may or may not the size of the ventricle. • In hypertension, • valvularstenosis,
eccentric hypertrophy • characterized by an increase in heart size as well as an increase in wall thickness. • In volume overload states (e.g., valvular regurgitation or abnormal shunts)
However, with sustained or worsening heart function, pathologic changes may eventually supervene, resulting in structural and functional disturbances; such degenerative changes include myocyte apoptosis, • cytoskeletal alterations, • and altered extracellular matrix synthesis and remodeling. • Even hypertrophy comes at a significant
. Oxygen requirements of the hypertrophic myocardium are increased as a result of increased myocardial cell mass and increased tension of the ventricular wall • the myocardium becomes vulnerable to ischemic injury.
The most common causes of left-sided cardiac failure are • (1) IHD, • (2) systemic hypertension, • (3) mitral or aortic valve disease, • (4) primary diseases of the myocardium.
LVF is divided on clinical grounds into • Systolic Failure (pump failure) • Diastolic failure • There is stiffening of the left ventricle biut output is maintained at rest.but during exercise there is increase in filling pressure leads to backflow of pressure into pulmonary circulation causing Pulmonary edema
It is commonly seen after 65yers and more common in women. • Constrictive pericardiits • Amyloidosis • Restrictive cardiomyopathy • Myocardial fibrosis • Also in elderly persons due to age stiffening common.
The most common cause of right-sided heart failure is • 1. left ventricular failure, with its associated pulmonary congestion and elevation in pulmonary arterial pressure. • Right-sided failure can also occur in the absence of left-sided heart failure in patients with intrinsic 1.diseases of the lung parenchyma and/or pulmonary vasculature (corpulmonale) • 2.primary pulmonic or tricuspid valve disease.
Left Heart Failure Symptoms • Dyspnea • on exertion • at rest • Orthopnea • redistribution of peripheral edema fluid • graded by number of pillows needed • Paroxysmal Nocturnal Dyspnea (PND)
LEFT Heart Failure Dyspnea Orthopnea PND (Paroxysmal Nocturnal Dyspnea) Blood tinged sputum Cyanosis Elevated pulmonary “WEDGE” pressure (PCWP) (nl = 2-15 mm Hg)
Morphology of LVF • the left ventricle is usually hypertrophied and often dilated, sometimes quite massively. There are usually nonspecific changes of hypertrophy and fibrosis in the myocardium. Secondary enlargement of the left atrium with resultant atrial fibrillation
can reduce stroke volume or lead to blood stasis and thrombus formation (particularly in the atrial appendage); a fibrillating left atrium carries a substantially increased risk of embolic stroke • Rising pressure in the pulmonary veins is ultimately transmitted retrogradely to the capillaries, resulting in pulmonary congestion and edema. The lungs are heavy and boggy, and histologically there are perivascular and interstitial transudate, alveolar septal edema, and intra-alveolar edema
Heart failure cells. • Right-Sided Heart Failure • Liver and Portal System. The liver is usually increased in size and weight (congestive hepatomegaly), nutmeg liver congested red centers of the liver lobules are surrounded by paler, sometimes fatty, peripheral regions. • the central areas can become fibrotic, creating so-called cardiac cirrhosis.
Pleural and Pericardial Spaces • Subcutaneous Tissues. Peripheral edema of dependent portions of the body, especially ankle (pedal) and pretibial edema, is a hallmark of right-sided heart failure. In chronically bedridden patients, the edema may be primarily presacral. Generalized massive edema is called anasarca.
Right Sided Heart Failure • Etiology • left heart failure • cor pulmonale • Symptoms and signs • Liver and spleen • passive congestion (nutmeg liver) • congestive spleenomegaly • ascites • Kidneys • Pleura/Pericardium • pleural and pericardial effusions • transudates • Peripheral tissues
RIGHT Heart Failure FATIGUE “Dependent” edema JVD Hepatomegaly (congestion) ASCITES, PLEURAL EFFUSION GI Cyanosis Increased peripheral venous pressure (CVP) (nl = 2-6 mm Hg)
HEART DISEASE • CONGENITAL (CHD) • ISCHEMIC (IHD) • HYPERTENSIVE (HHD) • VALVULAR (VHD) • MYOPATHIC (MHD)
HEART DISEASE • CONGENITAL (CHD) • ISCHEMIC (IHD) • HYPERTENSIVE (HHD) • VALVULAR (VHD) • MYOPATHIC (MHD)
Etiology and pathogenesis The main cause is sporadic genetic abnormalities Single gene muation chromosomal deletion or additions Single gene mutations that affect proteins with signaling pathways
Mutations in NOTCH pathway associated with bicuspid aortic valve and TOF • Fibrillin mutation Marfan syndrome • The most common fgenetic cause is Trisomy 21. 40% of them have one or the other CHD
The envirnomental factors responsible for CHD are • Congenital rubella infection • Gestational diabtics • Teratogenic effects • And nutritional defect
CONGENITAL HEARTDEFECTS • Faulty embryogenesis (week 3-8) • Usually MONO-morphic (i.e., SINGLE lesion) (ASD, VSD, hypo-RV, hypo-LV) • May not be evident until adult life (Coarctation, ASD) • Overall incidence 1% of USA births • INCREASED simple early detection via non invasive methods, e.g., US, MRI, CT, etc.
The three main classification are • Malformations causing Left to right shunt • 1.ASD • 2.Patent formenovale • 3.VSD • 4.PDA • 5.Atrio ventricular septal defect
Malformation causing right to left shunt • 1.TOF • 2.Transpositions of great arteries • 3.Perstitent Trancusarteriosus • 4.Triscupid atresia
Obstructive congentialanomolies • Coarctation of aorta • PS and atresia • AS
ASD • It is an abnormal opening in the atrial septum due to incomplete tissue formation. • Usually asymptomatic until adulthood • SECUNDUM (90%): Defective fossaovalis • PRIMUM (5%): Next to AV valves, mitral cleft • SINUS VENOSUS (5%): Next to SVC with anomalous pulmonary veins draining to SVC or RA
CF • A murmer due to increaase flow of blood thr pulmonary valves. • Uncommon PH • Surgical closure.
Patent formenovalehole by tissue flap in the atrial septum. • Important during fetal circulation rt to left shunt from the placenta. • Closes spontaneously at birth remain open leads to PFO
VSD • By far, most common CHD defect • Only 30% are isolated • Often with TETRALOGY of FALLOT • 90% involve the membranous septum • If muscular septum is involved, likely to have multiple holes • SMALL ones often close spontaneously • LARGE ones progress to pulmonary hypertension