Boiling and Freezing
Boiling and Freezing. Chapter 14. Boiling. The process in which a liquid changes phase to a gas. Boiling Point. The temperature at which, under “Standard” conditions, a liquid will change into a gas. Boiling Point.

Boiling and Freezing
E N D
Presentation Transcript
Boiling and Freezing Chapter 14
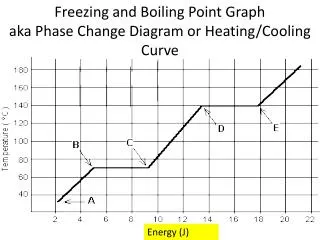
Boiling • The process in which a liquid changes phase to a gas.
Boiling Point • The temperature at which, under “Standard” conditions, a liquid will change into a gas.
Boiling Point • At the boiling point, the vapor pressure of the liquid will equal the outside atmospheric pressure. • No longer will the atmosphere “Hold” the liquid down (or in) and the liquid “Jumps” out of the container as a gas.
Boiling Point • The vapor pressure is determined by the number of solute particles, and the colligative properties of the solution.
Boiling Point • To increase the boiling point, add more solute. • ΔTb = Kb * m
Boiling Point • ΔTb = Kb * m • ΔTb = elevation of BP • Kb = molal constant • m = molality
Boiling Point • Chart page 500 • Chart page 501
Freezing point depression • ΔTf= Kf* m
Freezing point depression • You can figure it out…
Freezing point depression • Chart page 502
Video • Ice-Ice Baby • https://www.youtube.com/watch?v=25SHX5Sj7ZA • Slip sliding away • https://www.youtube.com/watch?v=Wp2sqrfGSPI • ??? • https://www.youtube.com/watch?v=ST1fUaKIBMs