Chapter 10 Nucleotides and Nucleic Acids
Chapter 10 Nucleotides and Nucleic Acids. Outline. What are the structure and chemistry of nitrogenous bases ? What are nucleosides ? What are the structure and chemistry of nucleotides ? What are nucleic acids ? What are the different classes of nucleic Acids ?

Chapter 10 Nucleotides and Nucleic Acids
E N D
Presentation Transcript
Outline • What are the structure and chemistry of nitrogenous bases ? • What are nucleosides ? • What are the structure and chemistry of nucleotides ? • What are nucleic acids ? • What are the different classes of nucleic Acids ? • Are nucleic acids susceptible to hydrolysis ?
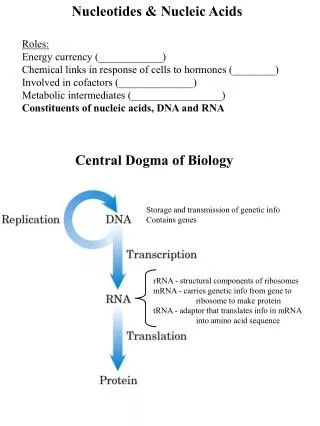
Information Transfer in Cells Central Dogma of Molecular Biology • Information encoded in a DNA molecule is transcribed via synthesis of an RNA molecule. • The sequence of the RNA molecule is "read" and is translated into the sequence of amino acids in a protein. • See Figure 10.1.
Information Transfer in Cells Figure 10.1 The fundamental process of information transfer in cells.
10.1 What Are the Structure and Chemistry of Nitrogenous Bases? Figure 10.2(a) The pyrimidine ring system; by convention, atoms are numbered as indicated. N1 is attached to ribose. (b) The purine ring system; atoms numbered as shown. N9 is attached to ribose.
10.1 What Are the Structure and Chemistry of Nitrogenous Bases? Know these structures Figure 10.3 The common pyrimidine bases – cytosine, uracil, and thymine – in the tautomeric forms predominant at pH 7.
10.1 What Are the Structure and Chemistry of Nitrogenous Bases? Know these structures Figure 10.4 The common purine bases – adenine and guanine – in the tautomeric forms predominant at pH 7.
10.1 What Are the Structure and Chemistry of Nitrogenous Bases? Figure 10.5 Other naturally occurring purine derivatives – hypoxanthine, xanthine, and uric acid.
The Properties of Pyrimidines and Purines Can Be Traced to Their Electron-Rich Nature • The aromaticity and electron-rich nature of pyrimidines and purines enable them to undergo keto-enol tautomerism. • The keto tautomers of uracil, thymine, and guanine predominate at pH 7. • By contrast, the enol form of cytosine predominates at pH 7. • Protonation states of the nitrogens determines whether they can serve as H-bond donors or acceptors. • Aromaticity also accounts for strong absorption of UV light at 260 nm. (Proteins absorb at 280 nm.)
The Properties of Pyrimidines and Purines Can Be Traced to Their Electron-Rich Nature Figure 10.6 The keto-enol tautomerism of uracil.
The Properties of Pyrimidines and Purines Can Be Traced to Their Electron-Rich Nature Figure 10.7 The tautomerization of the purine guanine.
The Properties of Pyrimidines and Purines Can Be Traced to Their Electron-Rich Nature Figure 10.8 The UV absorption spectra of the common ribonucleotides.
The Properties of Pyrimidines and Purines Can Be Traced to Their Electron-Rich Nature Figure 10.8 The UV absorption spectra of the common ribonucleotides.
10.2 What Are Nucleosides? Structures to Know • Nucleosides are formed when a base is linked to a sugar via a betaglycosidic bond. • The sugars are pentoses. • D-ribose (in RNA). • 2-deoxy-D-ribose (in DNA). • The difference - 2'-OH vs 2'-H. Primes are used in nucleosides and nucleotides but not sugars alone. • This difference affects secondary structure and stability.
10.2 What Are Nucleosides? Figure 10.9 The linear (Fischer) and cyclic (furanose) forms of ribose. Figure 10.9 The linear (Fischer) and cyclic (furanose) forms of deoxyribose.
10.2 What Are Nucleosides? • The base is linked to the sugar via a betaglycosidic bond. • The carbon of the glycosidic bond is anomeric. • Named by adding -idine to the root name of a pyrimidine or -osine to the root name of a purine. (Uracil uridine and adenine adenosine) • Conformation can be syn or anti. • Sugars make nucleosides more water-soluble than free bases.
10.2 What Are Nucleosides? Figure 10.10 The common ribonucleosides.
10.3 What Is the Structure and Chemistry of Nucleotides? • Nucleotides are nucleoside phosphates • Know the nomenclature. • "Nucleotide phosphate" is redundant! • Most nucleotides are ribonucleotides. • Nucleotides are polyprotic acids due to the phosphates.
10.3 What Is the Structure and Chemistry of Nucleotides? Figure 10.11 Structures of the four common ribonucleotides – AMP, GMP, CMP, and UMP. Also shown: 3’-AMP.
10.3 What Is the Structure and Chemistry of Nucleotides? Figure 10.13 Formation of ADP from AMP by the addition of a phosphate group forming a phosphoric anhydride linkage. Note that the reaction is a dehydration synthesis.
10.3 What Is the Structure and Chemistry of Nucleotides? Figure 10.13 Formation of ATP from ADP by the addition of a phosphate group forming a phosphoric anhydride linkage. This is also a dehydration process.
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy Nucleoside 5’-triphosphates are indispensable agents in metabolism because their phosphoric anhydride bonds are a source of chemical energy. Functions: • 1. ATP is central to energy metabolism: (see the following slides). • 2. Nucleotides serve as signal molecules and regulators: c-AMP and c-GMP. • 3. NTPs are substrates for DNA and RNA synthesis. The bases serve as recognition units.
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy Functions continued: • 4. Nucleotides are high energy carrier molecules: • GTP is involved in protein synthesis (translation). • Initiation, elongation and termination steps. • And ATP in activation: Aminoacyl-AMP. • CTP is involved in lipid synthesis. • CDP-diacylglycerol, etc. • UTP is involved in carbohydrate metabolism. • UDP-glucose, etc. • 5. Nucleotides are redox cofactors: • NAD+, NADP+, FMN, FAD, Coenzyme A.
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy Figure 10.14 Phosphoryl, pyrophosphoryl, and nucleotidyl group transfer, the major biochemical reactions of nucleotides. Phosphoryl group transfer is shown here. Pyrophosphoryl group transfer is shown here.
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy Nucleotidyl group transfer is shown here.
10.3 What Is the Structure and Chemistry of Nucleotides? Figure 10.12 The cyclic nucleotide cAMP. Figure 10.12 The cyclic nucleotide cGMP
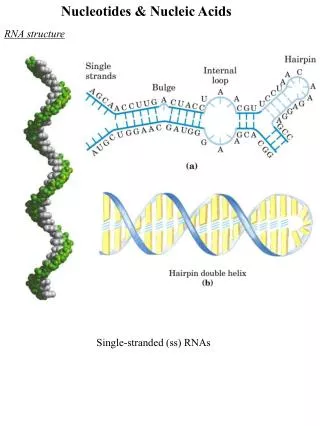
10.4 What Are Nucleic Acids? • Nucleic acids are linear polymers of nucleotides linked 3' to 5' by phosphodiester bonds. • Two types: • Ribonucleic acid and • Deoxyribonucleic acid. • Know the shorthand notations. • Sequence is always read 5' to 3', left to right. • In terms of genetic information, this corresponds to "N-terminal to C-terminal“ in proteins.
10.4 What Are Nucleic Acids? Figure 10.15 RNA 3',5'-phosphodiester bridges link nucleotides together to form polynucleotide chains. The 5'-ends of the chains are at the top; the 3'-ends are at the bottom.
10.4 What Are Nucleic Acids? Figure 10.15 DNA: 3’,5’-phosphodiester bridges link nucleotides together to form polynucleotide chains. The 5’-ends of the chains are at the top; the 3’-ends are at the bottom.
10.4 What Are Nucleic Acids? Shorthand notation for DNA. The bases are at the top. The vertical line is the sugar numbered top to bottom. The 5' end is to the left and the 3' end to the right. a and b are cleavage sites for nucleases. Linkage is a phosphodiester bond, each P has a (-) charge.
10.5 What Are the Different Classes of Nucleic Acids? • DNA - one type, one purpose (three forms). • RNA - Several types, several purposes: • ribosomal RNA - the basis of structure and function of ribosomes (largest amount). • messenger RNA - carries the message for protein synthesis (fewest and unique). • transfer RNA - carries the amino acids for protein synthesis (smallest molecules). • Others: • Small nuclear RNA. • Small non-coding RNAs. • Viral
10.5 What Are the Different Classes of Nucleic Acids? Figure 10.16 The antiparallel nature of the DNA double helix.
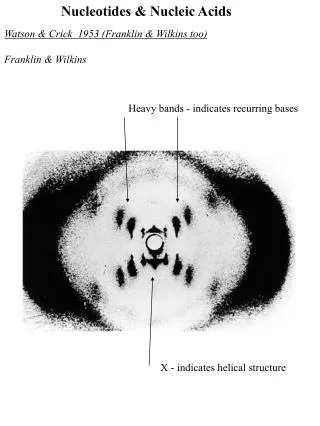
The DNA Double Helix • The double helix is stabilized by hydrogen bonds and hydrophobic interactions • "Base pairs" arise from hydrogen bonds. • Erwin Chargaff had the pairing data, but didn't understand its implications. • Rosalind Franklin's X-ray fiber diffraction data was crucial. • Francis Crick showed that it was a helix. • James Watson figured out the H bonds. • The hydrophobic effect from stacking of aromatic bases is also important.
The Base Pairs Postulated by Watson Figure 10.17 The Watson-Crick base pairing in A:T. Practice drawing this structure
The Base Pairs Postulated by Watson Figure 10.17 The Watson-Crick base pairing in G:C. Practice drawing this structure.
The Structure of DNA • An antiparallel double helix • Has a diameter of 2 nm. • Has a length of 1.6 million nm in E. coli. • Compact and folded (E. coli cell is only 2000 nm long). • Eukaryotic DNA is wrapped around histone proteins to form nucleosomes. • Base pairs: A-T, G-C.
Messenger RNA Carries the Sequence Information for Synthesis of a Protein Transcription product of DNA • In prokaryotes, a single mRNA contains the information for synthesis of many proteins • In eukaryotes, a single mRNA codes for just one protein, but structure is composed of introns and exons • See following slides.
Messenger RNA Carries the Sequence Information for Synthesis of a Protein Figure 10.20 Transcription and translation of mRNA molecules in prokaryotic versus eukaryotic cells. In prokaryotes, a single mRNA molecule may contain the information for the synthesis of several polypeptide chains within its nucleotide sequence.
Messenger RNA Carries the Sequence Information for Synthesis of a Protein In eukaryotics, mRNAs encode only one polypeptide but are more complex.
Eukaryotic mRNA • In eucaryotes, DNA is transcribed to produce heterogeneous nuclear RNA: • mixed introns and exons with poly A. • intron - intervening sequence. • exon - coding sequence. • poly A tail – stability ? • Splicing produces final mRNA without introns.
Ribosomal RNA Provides the Structural and Functional Foundation for Ribosomes • Ribosomes are about 2/3 RNA, 1/3 protein. • rRNA serves as a scaffold for ribosomal proteins. • The different species of rRNA are referred to according to their sedimentation coefficients. • rRNAs typically contain certain modified nucleotides, including pseudouridine and ribothymidylic acid. • The role of ribosomes in biosynthesis of proteins is treated in detail in Chapter 30. • Briefly: the genetic information in the nucleotide sequence of mRNA is translated into the amino acid sequence of a polypeptide chain by the ribosomes.
Ribosomal RNA Provides the Structural and Functional Foundation for Ribosomes Figure 10.21 Ribosomal RNA has a complex secondary structure due to many intrastrand H bonds. The gray line here traces a polynucleotide chain consisting of more than 1000 nucleotides. Aligned regions represent H-bonded complementary base sequences.
Ribosomal RNA Provides the Structural and Functional Foundation for Ribosomes Figure 10.22 The organization and composition of ribosomes.
Transfer RNAs Carry Amino Acids to Ribosomes for Use in Protein Synthesis • tRNAs are small polynucleotide chains. • 73 to 94 residues each, ~10% minor bases. • Several bases are usually methylated. • Each a.a. has at least one unique tRNA which carries the a.a. to the ribosome. • The 3'-terminus carries the amino acid and the 3'-terminal sequence is always CCA-a.a. • A tRNA with an amino acid attached is called an Aminoacyl tRNA. These molecules are the substrates for protein synthesis.
Ribosomal RNA Provides the Structural and Functional Foundation for Ribosomes Figure 10-23 Some unusual bases in DNA.
Transfer RNAs Carry Amino Acids to Ribosomes for Use in Protein Synthesis Figure 10.24 Transfer RNA also has a complex secondary structure due to many intrastrand hydrogen bonds. The black lines represent base-paired nucleotides in the sequence.
The Chemical Differences Between DNA and RNA Have Biological Significance • Two fundamental chemical differences distinguish DNA from RNA: • DNA contains 2-deoxyribose instead of ribose. • DNA contains thymine instead of uracil. Why does DNA contain thymine ? • Cytosine spontaneously deaminates to form uracil. • Repair enzymes recognize these "mutations" and replace these Us with Cs. • But how would the repair enzymes distinguish natural U from mutant U. • Nature solves this dilemma by using thymine (5-methyl-U) in place of uracil.
The Chemical Differences Between DNA and RNA Have Biological Significance Figure 10.26 The 5-methyl group on thymine labels it as a special kind of uracil. Figure 10.25 Deamination of cytosine forms uracil.
DNA & RNA Differences? Why is DNA 2'-deoxy and RNA is not? • Vicinal -OH groups (2' and 3') in RNA make it more susceptible to hydrolysis • DNA, lacking 2'-OH is more stable • This makes sense - the genetic material must be more stable • RNA is designed to be used and then broken down